All published articles of this journal are available on ScienceDirect.
Is Back-Projection Methodology Still Relevant for Estimating HIV Incidence from National Surveillance Data?
Abstract
Accurate estimates of HIV incidence are crucial to understand the extent of transmission of the infection, evaluate intervention strategies and effectively plan new public health control measures. HIV/AIDS surveillance systems in numerous industrialised countries record the number of known new HIV and/or AIDS diagnoses, which are often used as a surrogate marker for HIV incidence. HIV/AIDS diagnosis data have been used to reconstruct historical HIV incidence trends using modified back-projection methods. Estimates of HIV incidence are most robust when reliable data on the number of incident infections, a subset of all diagnoses, is widely available, and surveillance systems should prioritise the collection of these data. Back-projection alone provides reliable estimates of HIV incidence in the past, but is not useful when estimating current or future HIV incidence. However, back-projection methodology should be used in conjunction with other corroborative methods to estimate current HIV incidence, and methods to combine the various techniques should be investigated.
INTRODUCTION
Public health surveillance data have revealed an increase in HIV diagnoses in Australia and other industrialised countries over the last decade, predominantly among sub-populations of men who have sex with men (MSM) [1]. The temporal and geographical coverage of routinely collected data varies substantially between jurisdictions in the developed world [2]. Surveillance systems vary between and within countries depending on resource availability and existent infrastructure. Some surveillance activities are based on serial cross-sectional surveys to provide indicators about trends in HIV prevalence and/or underlying risk factors. The numbers of confirmed HIV and/or AIDS diagnoses are often maintained in provincial and national registries as another surveillance mechanism [3]. Such registries have become the core HIV surveillance data in numerous industrialised countries. Typically, these registries record the number of known new HIV diagnoses, and this is often interpreted as a proxy for HIV incidence. Accurate estimates of HIV incidence are difficult to obtain, due to the complex time lag between HIV infection and diagnosis [4]. Patterns in the numbers of diagnosed cases thus reflect the historical trajectory of the HIV epidemic and the rate of HIV testing [5], rather than the current rate of infection. Accurate HIV incidence estimates are required to guide the planning and evaluation of interventions for minimising transmission, and for the assessment of future health care needs [4-5]. Knowledge of the number of undiagnosed HIV infections is also important for understanding the extent of the epidemic and can be inferred from the relationship between prevalence and incidence estimates.
There are three broad methods to estimate HIV incidence: biological assays (such as the BED assay), mathematical modelling, and a statistical approach called back-projection (also known as back-calculation). These are the subject of current methodological research, and each method has different strengths and limitations. A review of biological assays and mathematical modelling was recently conducted by Hallett [6], with reference to these two methods as ‘two of the main approaches’ to estimating HIV incidence. However, back-projection is a also popular methodology that has been used to estimate HIV incidence and the number of undiagnosed infections [7] in Australia [8-13], the USA [4, 14-18], Canada [19], Switzerland [20], France/Germany/the UK [21], Thailand [22], the Caribbean [23], Malaysia [24], and Hong Kong [25]. In this article, the utility and relevance of back-projection methodology to estimate HIV incidence from diagnosis data is discussed in light of recent advances in mathematical modelling and biological assays, using the Australian HIV/AIDS surveillance system as an example.
Back-Projection Methodologies
Historically, back-projection was a widely used statistical method to estimate HIV incidence from data on AIDS diagnoses by assuming an incubation period from HIV infection to the onset of AIDS [16]. However, the introduction of antiretroviral therapy in the mid 1990s has resulted in a significant increase in the length of time between HIV infection and AIDS onset; indeed, under regular monitoring and effective therapies AIDS should not occur for people living with HIV. A far more complex pattern of incubation from HIV infection to clinical markers is very difficult to model due to variety in treatment regimens and their effect [9]. Several methods have been developed to ameliorate the uncertain duration of time between HIV acquisition and AIDS onset, including attempts to model the effect of anti-retroviral therapy (ART) [22], and the inclusion of CD4+ count information [20]. Inclusion of these variables enables models to more closely fit the true time lag observed between HIV infection and AIDS diagnosis; however, the models do not fully capture the complexity of the treatment regimens available or the very long time lag. Modified back-projection approaches that aim to estimate HIV incidence from the time between HIV acquisition and HIV diagnosis (rather than AIDS diagnosis) have been developed [9-10], and are reviewed elsewhere [26]. These methods allow the time lag to be modelled without the difficulties caused by ART, since therapy begins after HIV diagnosis. Modified back-projection methods reflect the decreasing utility of AIDS diagnosis as a surveillance tool in the developed world, and the shift away from the reporting of AIDS diagnoses in some countries, such as Australia. As such, the importance of newly diagnosed cases of HIV has increased. Further, identification of the subset of all newly diagnosed cases that can be classified as newly acquired infections provides information which can potentially be utilised for calculating incidence trends. The ‘Ottawa/Sydney’ method is a modified back-projection method which incorporates data on the number of recently acquired infections [26]. Several studies have been conducted to estimate HIV incidence in Australia using this back-projection methodology from the national surveillance data [9-10, 13].
Briefly, the ‘Ottawa/Sydney’ method reconstructs the historic HIV incidence curve using a formulation of the time between HIV acquisition and HIV diagnosis [27]. For this method, the frequency of HIV testing is estimated based on two testing forces. Firstly, it is assumed that a proportion of individuals with HIV infection are tested during asymptomatic infection, and are hence diagnosed prior to the onset of clinical symptoms (testing force 1). Secondly, it is assumed that other individuals seek testing due to the onset of clinical symptoms at a late stage of HIV progression (testing force 2), resulting in progression from HIV acquisition to diagnosis as in the absence of treatment. The HIV incidence curve is reconstructed by combining the models for testing forces 1and 2, using a mixing function based on the annual number of observed AIDS diagnoses (up to 1994, prior to which effective ART was not available), HIV diagnoses and recently acquired HIV infections.
The Utility of Back-Projection to Estimate Incidence from Surveillance Data
Useful insights into the Australian HIV epidemic have been gained using the ‘Ottawa/Sydney’ back-projection method to estimate HIV incidence. Australia has a comprehensive HIV surveillance system conducted by the Kirby Institute, in collaboration with the Australian Government Department of Health and Ageing, State and Territory health authorities and other networks [28]. Diagnoses have been notified and recorded for AIDS cases since 1981 and HIV cases since 1985. Information on cases of recently acquired HIV infection (as defined by a recent seroconversion illness or a previous negative test within 12 months) has been collected since 1991. State and territory health authorities forward newly diagnosed cases of HIV infection with information on demographic and clinical characteristics (including CD4+ cell count), and the most likely route of exposure to HIV, to the National HIV Registry [29].
The historical HIV incidence curve for Australian heterosexual and homosexual people, reconstructed by back-projection, shows that cumulatively by 2007 approximately 20,000 MSM were infected with HIV, of whom 13% had not yet been diagnosed [9]. Additionally, the mean age at the time of HIV infection has increased over the last decade [10]. Despite these important findings, there are limitations of the modified back-projection method for estimating HIV incidence. Knowledge of current HIV incidence is most useful for public health evaluation and planning, however, estimates of incidence in recent years are less stable than longer term trends [7]. Many recent infections will not yet have been diagnosed, and data in the most recent period of time will have the widest margin of error, as demonstrated in Fig. (1). In particular, HIV incidence estimates from the most recent three years can be highly unstable. The back-projection method does not allow for variation in testing rates, such as among individuals who seek testing following a high risk incident. Variability in testing rates is a critical variable that determines how far incidence estimates deviate from recorded prevalence. More explicit incorporation of testing rates into the model is required to accurately estimate incidence. The proportion of individuals allocated to each of the two testing force models, and hence which testing rate ‘profile’ they are assigned to in any year, depends on the accuracy of the data as to whether the case is newly acquired. It is likely that the number of newly acquired infections is underestimated in Australia, and in any other setting in which the Ottawa/Sydney method is applied. Diagnoses of newly acquired HIV infection are dependent on frequent HIV antibody testing and so they provide a lower bound to the extent of recent HIV transmission. Additionally, seroconversion illness, one of the criteria for the establishment of a newly acquired infection, may be mis-attributed.
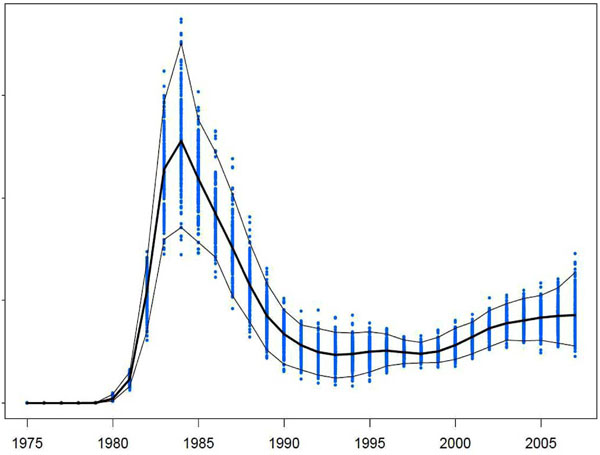
Estimated HIV incidence among men who have sex with men in Australia (1975 – 2007) and associated 95% confidence intervals.
Recommendations for National Surveillance
Developed countries generally have concentrated epidemics among particular sub-populations, such as MSM. Some complementary HIV surveillance activities include the periodic cross-sectional collection of biological data to estimate prevalence, and behavioural data to understand risk factors and to increase explanatory power among the affected sub-populations [3]. However, accurate estimates of HIV incidence are most desired in public health surveillance in order to assess the state of the HIV epidemic. Despite the limitations of back-projection methods, are they still relevant for estimating HIV incidence from surveillance data? Mathematical modelling approaches suffer from similar limitations to back-projection, and there is often a high degree of uncertainty in estimates from the most recent 5 years and in future epidemic trajectories [6]. These models are also highly dependent on the availability and quality of data used to derive model parameters. Alternatively, BED assays cannot determine past incidence, and despite improvements currently have a very high ‘false-recent’ rate [6]. While back-projection methods have been used extensively to reconstruct HIV incidence curves, there are no currently available data which establish the reliability of back-projection when compared to mathematical modelling and biological assays; nor are any comparative data available between different back-projection algorithms. Therefore, back-projection is one of a series of valid methods which are used to estimate HIV incidence from surveillance data, and further research is needed to determine the most reliable method.
HIV surveillance systems should place a high degree of importance on collecting data in such a way that enables good estimates of HIV incidence to accurately track the progress of the epidemic. The best results can be obtained within a public health framework that encourages frequent and widespread HIV testing, and that has consistent and reliable procedures in place across the diagnostic practitioner network to detect recently acquired infections. The back-projection method is unable to provide any projections for the future trajectory of the HIV epidemic, and is unreliable in the recent past. Previous literature has recommended that in order to obtain accurate estimates of HIV incidence in the very recent past, a ‘triangulation’ of data and methods should be considered [5]. Alternate methods of estimating HIV incidence, such as BED assays [30], mathematical modelling and cohort studies should be used in addition to back-projection [5], with consideration for the strengths and weaknesses of the various methods. Biological or statistical methods to improve the utility of the BED assay to measure HIV incidence is of particular importance [30], though at the current time it remains expensive and is not sufficiently accurate [31]. Back-projection methodology, however, does not require laboratory testing to determine newly acquired infections, or linkage to individual patient data. There have been no recommendations produced to date detailing how to combine evidence across multiple methodologies. Further research should focus on statistical and mathematical methods to combine the various approaches. Guidelines should be produced with recommendations for how to rank the various methods, and the degree to which they should corroborate to achieve a satisfactory result.
CONCLUSION
National surveillance data can be used to reconstruct historical HIV incidence trends using modified back-projection methods, such as the ‘Ottawa/Sydney’ method. However, back-projection is less reliable and relevant for estimating current trends and cannot be used to predict future trends. It is recommended that national surveillance systems emphasise accurate recording of recently acquired infections and the use of laboratory tests for incident HIV infection within a surveillance program. Back-projection methodology should be used in conjunction with other corroborative methods to estimate current HIV incidence.
ACKNOWLEDGEMENTS
The authors acknowledge funding from the Australian Government Department of Health and Ageing; and grant numbers FT0991990 and DP1093026 from the Australian Research Council. The views expressed in this publication do not necessarily represent the position of the Australian Government. The Kirby Institute is affiliated with the Faculty of Medicine, University of New South Wales.
CONFLICTS OF INTEREST
The authors confirm that this article content has no conflicts of interest.


