All published articles of this journal are available on ScienceDirect.
Recurrent Meningitis and Subarachnoid Hemorrhage Due to Salmonella in an HIV+ Patient: Case Report and Mini-Review of the Literature
Abstract
Meningitis due to non-typhi salmonella is infrequent in HIV-positive adults.
We report a case of a patient with >300 CD4+ cells/mm3 who presented with five episodes of recurrent meningitis, focal subarachnoid hemorrhage and cerebral vasculitis ultimately attributed to Salmonella choleraesuis infection. Even within the cART era invasive salmonellosis can occur in unusual ways in HIV-infected patients.
BACKGROUND
Recurrent invasive salmonellosis is accepted as an AIDS defining event in patients with HIV infection [1]. Meningitis due to Salmonella species is a relatively infrequent finding in adults. There is a paucity of published information regarding salmonellosis as the cause of cerebral vascular compromise with subarachnoid hemorrhage. We present a case of recurrent meningitis associated with subarachnoid hemorrhage in a patient with HIV disease.
CASE PRESENTATION
The patient is a 49 y.o male with a long history of HIV disease without prior opportunistic infections. He had a long history of partial responses and subsequent virologic failures to antiretroviral treatment. At the time of the presentation he was receiving tenofovir, stavudine, fosamprenavir and ritonavir, with a viral load ranging from 3.7 to 4.2 log copies in the last year and a most recent CD4+ T-cell count of 341 cells/mm3 (11.6%). The patient had been clinically stable until he presented with fever and meningismus of acute onset; with glucose in cerebrospinal fluid (SFC) of 12 mg%, 233 mg% of proteins and 975 leukocytes per mm3. Upon admission to the Critical Care Unit he was found to have a focal subarachnoid hemorrhage (SAH) in the area of the right middle cerebral artery (Fig. 1, left side). This was associated with mild impairment of consciousness and left VI nerve compromise. Angiogram imaging results were negative for either arteriovenous malformation or aneurysm.
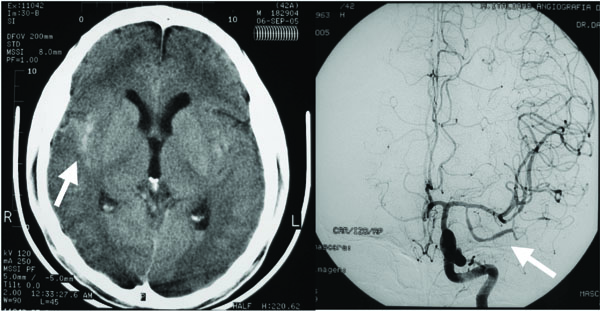
Left side: Computerized Tomography of brain showing focal Subarachnoid Hemorrhage in the right middle cerebral artery area (arrow). Right side: Angiogram showing vascular changes compatible with vasculitis (arrow).
Bacteriological, mycological and mycobacterial (including PCR) evaluations in the CSF remained negative, as a Non-treponemal syphilis test did.
Empiric antibiotic treatment with I.V. ceftriaxone was administered and the patient recovered uneventfully. He was discharged after one week. However, soon after discharge the patient had another acute episode of fever, meningeal signs and photophobia, with similar CSF findings (including CSF glucose of 4 mg%). An angiogram revealed multiple and bilateral short segments of narrowing and beading involving proximal and distal median cerebral vessels, with no evidence of arteriovenous malformation nor aneurysm (Fig. 1, right side). There was no evidence of recurrent cerebral bleeding and no microorganisms were detected. With the presumptive diagnosis of primary CNS vasculitis the patient was started on 1 mg/Kg of prednisone. Empiric coverage with ceftriaxone was also initiated but stopped after one week of negative cultures. He had a very rapid and complete clinical recovery and was discharged on oral steroids.
One week later the patient was hospitalized again due to a third episode of fever and meningeal signs. A lumbar puncture revealed a glucorrachia of 2 mg% and 1300 leukocytes per mm3. CSF cultures were negative. A CT scan and MRI showed no pathological findings and in a new angiogram the vascular findings were improved but still present. A meningeal biopsy was considered at that time but subsequently not performed due to a risk/benefit consideration, and the patient was continued on steroid therapy in addition to three weeks of empiric IV ceftriaxone therapy. Clinical recovery was rapid and complete, and CSF improvement was documented. Twelve days after antibiotic discontinuation the patient had a fourth similar episode with fever, marked impairment of consciousness and CSF findings continued to be consistent with meningitis. No organisms were detected and he received again an antibiotic course with complete response. The patient’s clinical and CSF statuses were evaluated one month after the beginning of the fourth episode; significant improvement was documented and his steroid therapy was discontinued.
During his last evaluation, an empiric course of high-dose IV penicillin was considered due to the rare possibility of seronegative neurosyphilis as the cause of recurrent aseptic meningitis masked by a partial response to ceftriaxone. However, within five days of the last examination the patient was admitted for his fifth recurrence of fever and meningismus. CSF characteristics were very similar to the previous episodes (glucose 2 mg%, proteins 446 mg%, leukocytes 140 per mm3), the CSF gram stain was negative, but bacteriologic culture detected the growth of a sensitive gram negative rod. This was identified as Salmonella species choleraesuis, identification subsequently confirmed at the Argentinian National Reference Center (Instituto Malbrán). This Salmonella isolate retained broad sensitivity to the common antimicrobials used to treat gram negative rod infection. On the basis of this new information the patient was treated with IV ceftriaxone for four weeks with complete remittance of the symptoms, and was subsequently placed on oral daily trimethoprim-sulfa-methoxazole (TMS) as permanent secondary prophylaxis. Of note, Salmonella was not detected in blood or fecal cultures. A summary of laboratory findings is shown in Table 1. A follow up angiogram showed complete reversion of the vascular findings.
Summary of Laboratory Tests Results
| CSF Parameters | 1st EpisodeDay 1 | 2nd EpisodeDay +17 | 3rd EpisodeDay +29 | Follow UpDay +34 | 4th EpisodeDay +62 | Follow UpDay +92 | 5th EpisodeDay +97 |
|---|---|---|---|---|---|---|---|
| Physical aspect | Bloody, Xantochromic | Turbid | Clean | Clean | Clean | Clean | Clean |
| Glucose (mg/dL) | 12 | 4 | 2 | 47 | 2 | 49 | 2 |
| CSF/Plasma Glucose | 0.09 | 0.03 | 0.02 | 0.51 | 0.015 | 0.015 | |
| Protein (mg/dL) | 233 | 374 | 298 | 64 | 537 | 34 | 446 |
| Leukocytes/mm3 | 975 | 197 | 1300 | 10 | 17 | 5 | 140 |
| % Neutrophils | 80 | 55 | 75 | 0 | 80 | 0 | 70 |
| % Lymphocytes | 20 | 45 | 25 | 0 | 20 | 0 | 30 |
| Red blood cells | 40.000 | 13.000 | 0 | 0 | 0 | 0 | 0 |
| Bacteriology | Neg | Neg | Neg | Neg | Neg | Neg | Salmonella |
| Bacterial Antigens | Neg | Neg | Neg | ||||
| Ziehl Neelsen | Neg | Neg | Neg | ||||
| TB culture | Neg | Neg | Neg | Neg | |||
| TB PCR | Neg | Neg | Neg | ||||
| HSV PCR | Neg | Neg | |||||
| VDRL | Neg | Neg | Neg | Neg | Neg | ||
| FTA Abs | Neg | Neg | Neg | ||||
| Fungal culture | Neg | Neg | Neg | Neg | Neg | Neg | |
| Crypto Antigen | Neg | Neg | |||||
| Lactic acid (mmol/L) | 9.5 | 10.8 | 10.5 | ||||
| Other parameters | |||||||
| Plasma Leukocytes/mm3 | 10.780 | 19.020 | 4.470 | 17.550 | |||
| Blood culture | Neg | Neg | Neg | Neg | Neg | ||
| Fecal culture | Neg | Neg |
Neg: Negative result.
Summary of Characteristics of Previously Reported Cases of NTS Meningitis in Adults with HIV-Infection
| Patient/Refs. | Age/ Sex | Presentation | SalmonellaStrain | CSF WBC/mm3 | CSF Protein (mg/dl) | CSF Glucose (mg/dl) | Outcome |
|---|---|---|---|---|---|---|---|
| 1 [7] | 34-F | Fever, meningismus and multi-organ failure | choleraesuis | 40 | 75 | ND | Died |
| 2 [8] | 34-F | Obtundation, fulminant illness | heideberg | 0 | 342 | ND | Died |
| 3 [8] | 42-M | Obtundation, fulminant illness | enteritidis | 24 | 472 | ND | Died |
| 4 [8] | 47-M | Relapsing meningitis, brain abscess | enteritidis | 925 (initial case) | 199 (initial case) | ND | Recovered |
| 5 [9] | 35-M | ND | enteritidis | 1,200 | 228 | 21 | Recovered |
| 6 [10] | 42-M | Obtundation, fever, mild meningeal signs | Group D (+Cryptococcus) | 826 | 420 | 13 | Recovered |
| 7 [5] | 34-F | ND | typhimurium | ND | ND | ND | Died |
| 8 [11] | 28-M | Fever, meningismus | infantis | 8,640 | 355 | 25 | Recovered |
| 9 [12] | 43-M | Fever, headache, malaise confusion | enteritidis | 38,000 | 150 | 8 | Died |
| 10 [13] | 40-M | Fever, diarrhea, malaise, thrush | typhimurium | 8,965 | 298 | 5 | Recovered |
ND: No data available.
CASE DISCUSSION AND REVIEW OF THE LITERATURE
Since the mid 1980s, recurrent invasive salmonellosis was interpreted as a clinical marker of AIDS based on reports of increased risk and higher mortality rates of bloodstream infections in patients with HIV disease [1-5]. Its relation with cellular immunodeficiency is also shown by the increased susceptibility to invasive infections of non-typhi species (NTS) of Salmonella observed in patients with hematologic malignancies, metastatic solid tumors or under immunosuppressive therapy [6]. Focal complications of NTS bacteremia, although thought to occur more frequently in HIV-infected patients than in the general population, are still an uncommon complication in adults. In a case series published by Fernandez Guerrero et al., 10 of 38 patients with salmonellosis had focal suppurative complications, with the urinary tract and lungs being the most frequent sites of infection [7].
Meningitis due to NTS is a rare complication even in HIV-infected patients, affecting mainly infants and associated with a high mortality rate. A systematic search through MEDLINE, EMBASE and LILACS revealed that only few cases of NTS meningitis in adults have been published, even during the pre-HAART era [5,7-13]. The presentation and findings of the published cases are summarized in Table 2. Clinical manifestations appear to be typical for bacterial meningitis, and although CSF white blood cell counts have varied widely, elevated proteins and decreased CSF glucose levels were common findings in the reported cases. The mortality rate associated with NTS focal complications is high, estimated in the order of 30% in published series, and particularly in the case of meningitis it may be close to 50% [7]. Impairment of consciousness at presentation has been described as a risk factor for mortality in such cases [11]. The rapid initiation of adequate empiric therapy in the case described above, coupled with preservation of antibiotic sensitivity in the invading organism, may have accounted for the favorable outcome of the five clinical episodes.
Even though the risk of endovascular infection secondary to Salmonella bacteremia is well recognized and every arterial site may potentially become compromised with mycotic aneurisms, to our knowledge this is the first reported case of a NTS infection localized within the central nervous system presenting as SAH in an HIV positive adult [14-16].
It is likely that high-grade bacteremia is required for meningeal invasion, although in the case of NTS this bacteremia may have not been detected in up to 40% of reported cases [11]. More surprising in this case was the absence of growth of the Salmonella isolate in the CSF in the four initial episodes, suggesting either an extremely low organism burden at the time of sampling or a lack of communication between the initial site of CNS invasion and the CSF during those earlier episodes. Of note, since this patient had more than 200 CD4+ cells/mm3, he had not been receiving prophylaxis for opportunistic infections with TMS. Only one of the previously published cases of NTS meningitis appeared in the context of prophylaxis with TMS [11].
Our patient was started on daily prophylaxis after the last course of ceftriaxone and has had no relapses after more than two years of follow up.
Different types of vasculitides have been associated with HIV disease, although it is not always clear if the former is a consequence of the latter or merely represents an unfortunate coincidence [17,18]. Nevertheless, Salmonella species as well as other infectious agents have been previously postulated to be a cause of secondary vasculitis in patients with HIV infection either through direct microbial invasion or immune-mediated injury [19]. It is also known that certain other infectious processes such as tuberculosis, varicella zoster or fungal infections may cause intracranial hemorrhage, yet no association with invasive salmonellosis has been previously described [20,21]. Although a CNS biopsy could not be performed, the characteristics revealed by the imaging studies of bilateral, multiple, medium size arterial involvement, affecting both proximal and distal branches coupled with the temporal response to steroid therapy seen in this case, favored the diagnosis of secondary vasculitis over that of prolonged vasospasm or a single occult mycotic aneurysm.
CONCLUSIONS
In summary, we describe here an unusual case of recurrent meningitis due to Salmonella choleraesuis in an HIV-infected patient presenting as a focal subarachnoid hemorrhage associated with probable neurovasculitis who responded adequately to antimicrobial and steroid therapy.
Salmonella should be considered as a potential cause of recurrent, refractory meningitis in HIV patients, and should be included in the differential diagnosis even if a clear bacterial cause is not immediately identified. Even within the cART era invasive salmonellosis can still be a cause of clinical deterioration in patients with moderately advanced HIV disease, in particular in those who are not receiving routine prophylaxis with TMS.
CONSENT
Written informed consent was obtained from the patient for this publication and a copy (in Spanish) of this document is available for review by the Editor-in-Chief of this journal.
COMPETING INTERESTS
Authors declare no competing interests, either financial or non-financial.
No financial support was received for the preparation of this manuscript.
AUTHORS´ CONTRIBUTIONS
WHB, MR and AGP contributed with the development of the idea, data collection and the process of writing of the manuscript draft
GSG and MRA contributed with the analysis and data collection of the case
RTD, MLS and IMO participated in the correction of the final version of the present manuscript
ACKNOWLEDGEMENTS
We thankfully acknowledge Prof. Giuseppe Barbaro, Dr. Roberto Arduino, and Dr. Michael Sneller for their helpful thoughts in the management of this case and Virginia Garrote, María Laura Arias and Geraldina Surra for their help in the preparation of this manuscript.


