All published articles of this journal are available on ScienceDirect.
A Model and Risk Score for Predicting Nevirapine-Associated Rash among HIV-infected Patients: In Settings of Low CD4 Cell Counts and Resource Limitation§
Abstract
Background:
Rash is the most common adverse effect associated with nevirapine (NVP). We aimed to develop a model and risk score for predicting NVP-associated rash among HIV-infected patients with low CD4 cell counts.
Methods:
Cross-sectional study was conducted and 383 HIV-infected patients consecutively enrolled in the study.
Results:
Of 222 patients in the training set, 116 (52.2%) were males and median (IQR) age was 35.2 (31.1-42.0) years. Median (IQR) CD4 cell count was 104 (35-225) cells/mm3. Of these, 72 and 150 patients were in “rash” and “no rash” group, respectively. Four factors were independently associated with rash: a history of drug allergy (odds ratio (OR) 4.01, 95% confidence interval (CI), 1.75-9.20, P = 0.001), body weight <55 kg. (OR 2.02, 95% CI, 1.09-3.76, p = 0.026), not receiving slow dose escalation (OR 2.00, 95% CI, 1.06-3.77, p = 0.032), and no concomitant drug(s) (OR 2.48, 95% CI, 1.32-4.64, p = 0.005). Receiver-operator characteristic analysis yielded area under the curve of 71% and the goodness-offit statistics was 6.48 (p = 0.840). The variables were given scores of 14, 7, 7 and 9, respectively. A cutoff >21 points defined the high risk individuals which yielded specificity and positive predictive value of 99% and 69%, respectively, with OR of 3.96 (95% CI, 1.79-8.86, p = 0.001).
Conclusions:
A model and risk score for predicting NVP-associated rash performed well in this study population. It might be useful for predicting the risk of rash before NVP initiation among HIV-infected patients with low CD4 cell counts.
INTRODUCTION
Some factors including patients’co-morbidity, adherence, convenience, adverse drug effects, drug-drug interactions, and pregnancy potential have to be considered when selecting an initial regimen of antiretroviral therapy (ART) for HIV-infected patients [1]. In addition, one should consider gender and pretreatment CD4 cell counts in case of nevirapine (NVP) initiation [1]. Use of non-nucleoside reverse transcriptase inhibitor-based regimens as initial therapy can preserve protease inhibitors (PIs) for later use, thus reducing or delaying patient exposure to some of the adverse effects of PIs [1].
NVP is widely used as a component of the first-line ART in resource-limited countries because of the availability of generic drug at low cost and fixed-dose combination tablets. Nevertheless, NVP is not recommended to use in adult females with CD4 >250 cells/mm3 and males with CD4 >400 cells/mm3 due to a high rate of hepatotoxicity [1]. Furthermore, the development of rash in HIV-infected individuals receiving NVP is the most common adverse effect especially during the first few weeks of therapy, with frequencies ranging from 9 to 32% [2-8]. Rare but serious hypersensitivity reactions, Stevens-Johnson Syndrome and toxic epidermal necrolysis, had been reported [5, 9, 10]. Risk factors for NVP-associated rash were female gender including pregnant women [11-14], lower body weight [15], initiation of NVP at high CD4 cell counts [11, 12, 15, 16], pretreated with antiretroviral drugs less than 12 months [17], higher plasma NVP level [17], and genetic (human leukocyte antigen) [18, 19]. However, some factors may not be applicable to patients who were initiated NVP at low CD4 cell counts in resource-limited settings. In addition, some risk factors differ among study population and from patients to patients.
NVP-associated rash can be minimized by escalating the initial dose of NVP or lead-in prescription [13, 20]. Despite this intervention, rash continues to be the leading cause of NVP discontinuation [21, 22]. Use of antihistamines and/or glucocorticoids cannot prevent this adverse effect [13, 23, 24]. In some situations, NVP-based regimen is the only regimen that can be used, but one cannot predict the probability or risk of NVP-associated rash before NVP initiation for each particular individuals. We aimed to develop a model and risk score for predicting NVP-associated rash among HIV-infected patients who were initiated NVP at low CD4 cell counts in resource-limited settings.
METHODS
A cross-sectional study of HIV-infected patients, who attended the Infectious Diseases Clinic, Ramathibodi Hospital (a 1,000-bed University Hospital), Mahidol University, Bangkok, Thailand was conducted. Consecutive enrollment of patients had been done between March 2006 and August 2007. Inclusion criteria were adult (>15 years old) HIV-infected patients and received a generic fixed-dose combination of stavudine, lamivudine, and NVP (GPO-VIR®). Each eligible patient was followed, at least, from initiating NVP until six weeks after NVP initiation. Rash associated with NVP was diagnosed by physicians. If patients received concomitant drugs, which also had a high possibility to cause rash and the diagnosis of causative drug was equivocal, these patients were not included in the study. Concomitant drugs defined as any drugs apart from antiretroviral drugs of the current regimen, i.e., co-trimoxazole, fluconazole, anti-tuberculosis drugs, antihypertensive drugs, and lipid-lowering agents.
To obtain an unbiased estimate of sample population, 60% of study population was randomly selected from a total study population as a training set. The rest was used as a validation set. Demographic and clinical data were retrieved and reviewed retrospectively as previously described [15]. The study was approved by the Institutional Review Board.
Statistical Analysis
Continuous variables with normal distribution are presented as mean and standard deviation (SD) and those with non-normal distribution are shown as median and interquartile range (IQR). Continuous variables were compared between groups using independent t-test or Mann-Whitney U test. Continuous variables were divided into categories to facilitate risk estimation according to the distribution of the variables as well as clinical significance. Age was categorized into ≤35 vs >35 years old, body weight was categorized into ≤55 vs >55 kg., and CD4 cell counts were categorized into ≤100 vs >100 cells/mm3. Categorical variables are shown as frequency and percentage. Chi-square test or Fisher’s exact test was used for analysis where appropriate for categorical ones.
Simple logistic regression analysis was used to determine the association between risk factors and outcome (presence of NVP-associated rash). Variables that presented p<0.25, were considered in a multiple logistic regression model after assessment of multicollinearity of variance inflation factors. We also considered two more variables, gender [11, 25-27] and previous AIDS-defining illness [15], that had been reported as the risk factors for NVP-associated rash in the model. Variables were selected into a multiple logistic regression model with backward selection. The model was reduced by excluding variables with p>0.05 in order to retain a simpler diagnostic model containing only the strongest determinants of NVP-associated skin rash. Thus, variables that attained a level of significance (p<0.05) were retained in the model [28]. We then compared the final model from backward selection with the initial model by using a likelihood ratio test and Akaike's information criterion. A weighted risk score was constructed using logistic regression coefficients. These coefficients were converted into scores by multiplying by ten and rounded off to the nearest whole number that were added up to obtain an aggregated score [29].
Two performance indexes were used to estimate the discrimination and calibration of the predictive model. The discrimination was evaluated using the area under the receiver-operator characteristic (ROC) curve [30] and the calibration was measured using the Pearson’s goodness-of-fit test. An area under the ROC curve of 1.0 is ideal whereas it is <0.5 indicates no diagnostic accuracy. In general, the area under the ROC curve >0.7 indicates a useful test [31]. The Pearson’s goodness-of-fit test has a p-value of >0.05 is considered being the good calibration model. The optimum cut-off point for the score was determined by sensitivity, specificity, positive predictive value, and negative predictive value. A p-value <0.05 was considered statistically significance. All statistical analyses were performed using Stata statistical software version 10.0 (Stata Statistical Software: Release 10.0, Stata Corporation, College Station, TX, 2007).
RESULTS
Initially, 383 patients were included in the study but 13 (3.4%) patients were excluded because of missing clinical data of interests. The derivation group from random sampling was further analyzed. A total of 222 patients with a median (IQR) age of 35.2 (31.1-42.0) years were in a training set. Of these, 116 (52.2%) were males and median (IQR) body weight was 54.0 (48.0-63.0) kg. The most common route of HIV acquisition was heterosexual (98%). Thirty-two (14.4%) patients had a history of drug allergy and 134 (60.4%) patients receiving concomitant drug(s). Median (IQR) CD4 cell count at time of NVP initiation was 104 (35-225) cells/mm3. Prior to NVP initiation, 154 (69.4%) patients were naive to ART. Slow dose escalation of NVP was prescribed in 151 (68.0%) patients.
There were 72 patients in “rash” group and 150 patients in “no rash” group. Of 72 patients, 34, 35, and 3 patients developed grade 2, 3, and 4 of rash according to Division of AIDS table for grading the severity of adult and pediatric adverse events [32]. In rash group, a higher proportion of male gender (52.8% vs 45.3%, p = 0.318), patients with a history of drug allergy (25.0% vs 9.3%, p = 0.004), and switching from efavirenz (EFV) (26.4% vs 16.7%, p = 0.106) were found. In contrast, patients without rash had a higher proportion of receiving concomitant drug(s) (66.7% vs 47.2%, p = 0.008), naive to ART (75.3% vs 56.9%, p = 0.008), and prescribed slow dose escalation of NVP (74.0% vs 55.6%, p = 0.009). Median CD4 cell count at the time of NVP initiation was higher among patients in rash group (191 vs 72 cells/mm3, p = 0.001). Baseline demographics and clinical characteristics of patients by NVP-associated rash status are shown in Table 1.
Baseline Demographics and Clinical Characteristics of Patients by Nevirapine-Associated Rash Status
| Variables | Total (n = 222) | Rash (n = 72) | No Rash (n = 150) | P-Value |
|---|---|---|---|---|
| Age, median (IQR), years | 35.2 (31.1-42.0) | 35.0 (31.2-40.6) | 35.2 (30.9-42.2) | 0.918 |
| Male, n (%) | 116 (52.2) | 38 (52.8) | 68 (45.3) | 0.318 |
| Body weight, median (IQR), kg. | 54.0 (48.0-63.0) | 53.0 (48.0-59.0) | 55.0 (48.0-65.0) | 0.918 |
| HIV transmission by heterosexual, n (%) | 217 (97.8) | 69 (95.8) | 148 (98.7) | 0.227 |
| History of AIDS-defining illness, n (%) | 107(48.2) | 31 (43.1) | 76 (50.7) | 0.317 |
| Concomitant other underlying illness(s), n (%) | 32 (14.4) | 13 (18.1) | 19 (12.7) | 0.310 |
| History of drug allergy, n (%) | 32 (14.4) | 18 (25.0) | 14 (9.3) | 0.004 |
| Receiving concomitant drug(s), n (%) | 134 (60.4) | 34 (47.2) | 100 (66.7) | 0.008 |
| Naive to antiretroviral therapy, n (%) | 154 (69.4) | 41 (56.9) | 113 (75.3) | 0.008 |
| Switching from efavirenz, n (%) | 44 (29.8) | 19 (26.4) | 25 (16.7) | 0.106 |
| Receiving slow dose escalation of NVP, n (%) | 151 (68.0) | 40 (55.6) | 111 (74.0) | 0.009 |
| CD4 cell count at time of NVP initiation, median (IQR), cells/mm3 | 104 (35-225) | 191 (61-290) | 72 (23-200) | 0.001 |
Abbreviation: IQR, interquartile range, NVP, nevirapine.
Simple Logistic Regression Analysis of the Variables Related to Nevirapine-associated Rash
| Variables | Odds Ratio | 95% CI | P-Value |
|---|---|---|---|
| Male gender | 1.35 | 0.77-2.37 | 0.299 |
| Age (>35 vs ≤35 years old) | 0.97 | 0.56-1.71 | 0.926 |
| Body weight (≤55 vs >55 kg.) | 1.72 | 0.97-3.07 | 0.065 |
| HIV transmission by heterosexual | 0.31 | 0.05-1.90 | 0.206 |
| History of AIDS-defining illness | 0.74 | 0.42-1.30 | 0.289 |
| Concomitant other underlying illness(s) | 1.52 | 0.70-3.28 | 0.287 |
| History of drug allergy | 3.24 | 1.50-6.97 | 0.003 |
| No concomitant drug(s) | 2.24 | 1.26-3.97 | 0.006 |
| Naive to antiretroviral therapy | 2.31 | 1.27-4.19 | 0.006 |
| Switching from efavirenz | 1.79 | 0.91-3.53 | 0.091 |
| Not receiving slow dose escalation of NVP | 2.28 | 1.26-4.11 | 0.006 |
| CD4 cell count at time of NVP initiation (>100 vs ≤100 cells/mm3) | 2.69 | 1.49-4.83 | 0.001 |
Abbreviation: CI, confidence interval, NVP, nevirapine.
Multiple Logistic Regression Analysis of the Variables Related to Nevirapine-Associated Rash
| Variables | Estimated Regression Coefficient (Standard Error) | Odds Ratio | 95% CI | P-Value | Score (Points) |
|---|---|---|---|---|---|
| History of drug allergy | 1.38 (0.42) | 4.01 | 1.75-9.20 | 0.001 | 14 |
| No concomitant drug(s) | 0.91 (0.32) | 2.48 | .132-4.64 | 0.005 | 9 |
| Body weight (≤55 vs >55 kg.) | 0.70 (0.32) | 2.02 | 1.09-3.76 | 0.026 | 7 |
| Not receiving slow dose escalation of NVP | 0.69 (0.32) | 2.00 | 1.06-3.77 | 0.032 | 7 |
Abbreviation: CI, confidence interval, NVP, nevirapine.
Distribution of HIV-Infected Patients with and without Nevirapine-Associated Rash in Relation to the Risk Score Obtained
| Risk Score | Rash n (%) | No Rash n (%) | Total n (%) |
|---|---|---|---|
| 0 | 3 (4.2) | 31 (20.7) | 34 (15.3) |
| 7 | 14 (19.4) | 50 (33.3) | 64 (28.8) |
| 9 | 4 (5.6) | 16 (10.8) | 20 (9.0) |
| 14 | 9 (12.4) | 14 (9.4) | 23 (10.4) |
| 16 | 20 (27.8) | 24 (16.0) | 44 (19.8) |
| 21 | 4 (5.6) | 2 (1.3) | 6 (2.7) |
| 23 | 9 (12.4) | 8 (5.3) | 17 (7.7) |
| 28 | 4 (5.6) | 3 (2.0) | 7 (3.2) |
| 30 | 4 (5.6) | 1 (0.7) | 5 (2.2) |
| 37 | 1 (1.4) | 1 (0.7) | 2 (0.9) |
| Total | 72 (100.0) | 150 (100.0) | 222 (100.0) |
The equation used for the NVP-associated rash risk score calculation: NVP-associated rash risk score = 14 (a history of drug allergy) + 9 (no concomitant drug(s)) + 7 (body weight ≤55 kg.) + 7 (not receiving slow dose escalation of NVP)
We found five factors related to NVP-associated rash by simple logistic regression. These were a history of drug allergy (odds ratio (OR) 3.24, 95% confidence interval (CI), 1.50-6.97, p = 0.003), no concomitant drug(s) (OR 2.24, 95% CI, 1.26-3.97, p = 0.006), naive to ART (OR 2.31, 95% CI, 1.27-4.19, p = 0.006), not receiving slow dose escalation of NVP (OR 2.28, 95% CI, 1.26-4.11, p = 0.006), and CD4 cell counts at time of NVP initiation >100 cells/mm3 (OR 2.69, 95% CI, 1.49-4.83, p = 0.001) (Table 2).
In multiple logistic regression, the independent predictor variables of NVP-associated rash are shown in Table 3. These were a history of drug allergy (OR 4.01, 95% CI, 1.75-9.20, p = 0.001), no concomitant drug(s) (OR 2.48, 95% CI, 1.32-4.64, p = 0.005), body weight ≤55 kg. (OR 2.02, 95% CI, 1.09-3.76, p = 0.026), and not receiving slow dose escalation of NVP (OR 2.00, 95% CI, 1.06-3.77, p = 0.032). A ROC curve was plotted to determine the ability of the model for correctly predicting the NVP-associated rash. The area under the ROC curve was 71%, indicating good model discriminatory power between individuals with and without rash (Fig. 1). The Pearson’s goodness-of-fit statistic value was 6.48 (p = 0.840), indicating excellent model calibration for the observed versus the predicted outcome. When the model was applied to the remaining 148 patients who comprised the validation set, the model still showed good calibration (Pearson’s goodness-of-fit statistic of 15.61, p = 0.156) and fair discrimination (area under the ROC curve of 65%).
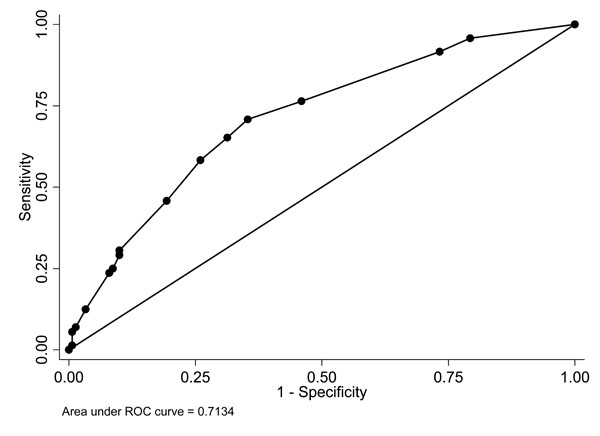
Receiver-operator characteristic (ROC) plot of the model composed of a history of drug allergy, body weight ≤55 kg., not receiving slow dose escalation, and no concomitant drug(s).
A numerical score was formulated by using logistic regression coefficients for risk score calculation. A history of drug allergy, body weight ≤55 kg., not receiving slow dose escalation of NVP, and no concomitant drug(s) were assigned scores of 14, 9, 7 and 7, respectively. The equation used for the NVP-associated rash risk score calculation for each patient was: NVP-associated rash risk score = 14 (a history of drug allergy) + 9 (no concomitant drug(s)) + 7 (body weight ≤55 kg.) + 7 (not receiving slow dose escalation of NVP). For each of the variables, a value of “1” was assigned if the variable was present or “0” of it was absent. The median (IQR) risk score of patients in rash group and no rash group was 16 (9-22) points and 7 (7-16) points, respectively (p<0.001). The distribution of risk score among HIV-infected patients with and without NVP-associated rash is shown in Table 4. If the total score was ≥21 points, the individual was defined as high risk for NVP-associated rash, otherwise was low risk. There were 22 of 72 (30.6%) patients in rash group and 15 of 150 (10.0%) had a risk score above the cutoff who were defined as high risk individuals (p<0.001). A cutoff at 21 points yielded specificity and positive predictive value of 99% and 69%, respectively. In addition, patients who had a risk score ≥21 points had odds of NVP-associated rash 3.96 times higher than those who had a score of <21 points (OR 3.96, 95% CI, 1.79-8.86, p = 0.001).
DISCUSSION
The present study was the first one, which was conducted to develop prediction model and risk score for NVP-associated rash in advanced HIV-infected patients. Approximately half of the patients had a history of AIDS-defining illness and the median CD4 cell count at time of NVP initiation was approximately 100 cells/mm3. This study well represented the target population in resource-constrained settings. According to clinical characteristics between the two groups, there was a lower proportion of patients prescribed slow dose escalation of NVP in rash group (55.6% vs 74.0%, p = 0.009). Furthermore, patients in rash group also had a higher proportion of switching EFV to NVP but there was no statistically significant difference (26.4% vs 16.7%). These two factors might be correlated because when changing from EFV to NVP, full dose of NVP should be started. Slow dose escalation of NVP after EFV treatment can lead to lower therapeutic plasma drug levels because EFV induces the metabolism of co-administered drugs through the induction of CYP 3A4 as it has been shown in a pharmacokinetic study [33]. However, multicollinearity was not found after variance inflation factors assessment of these 2 variables.
We found four independent factors associated with rash, namely a history of drug allergy, no concomitant drug(s), body weight ≤55 kg., and not receiving slow dose escalation. These factors had been reported as related factors for NVP-associated rash in the previous studies [13, 15, 17, 20, 34]. Patients who had a history of drug allergy had been reported as a risk factor of NVP-associated rash among Thai HIV-infected patients, especially those who had a history of sulfamethoxazole allergy [34]. Patients who took concomitant drug(s), which mostly (88.8%) were fluconazole and co-trimoxazole (data not shown), might have lower NVP plasma level than normal value due to enzyme induction of, especially, fluconazole [35]. The probable higher plasma NVP level might explain why patients who did not take concomitant drug(s) had a higher risk of rash. In addition, patients who were not taking other medications had a lower proportion of previous AIDS-defining illness (31.8% vs 59.0%, p<0.001) (data not shown) which reflected in higher CD4 cell counts. Both AIDS-defining illness and high CD4 cell counts at the time of NVP initiation were found to be associated with NVP-associated rash in the previous study [15]. Patients with higher body weight might have lower NVP plasma concentration and linked to lower risk of NVP-associated rash [36]. Clearly, slow dose escalation of NVP minimizes the risk for toxicity [13, 20].
No association between gender and CD4 cell count and risk of NVP-associated rash were observed in the present study even though these two factors were consistently reported in some studies [11-16]. We did not find any association between gender and risk of rash in our previous study using the same cohort [15]. CD4 cell count was associated with rash in simple logistic regression analysis of our present data, but it was not a risk factor after control of other variables.
Despite the fact that NVP-based regimen is the most commonly use in many countries, there was no model developed for health care providers to guide and/or predict NVP-associated rash before prescribing NVP especially in resource-limited settings. These factors from our model could be combined to create a risk index and risk score for predicting NVP-associated rash. Identifying risk factors associated with rash is relevant when health care provider need to prescribe NVP to HIV-infected patients. Counseling and informing patients regarding risk of rash associated with NVP is crucial for prevention the consequence from NVP adverse effects including adherence improvement [37]. This particular model was developed by selecting the significant factors from logistic regression analysis including factors that had an a priori biological and/or clinical rationale. A weighted risk score was constructed using logistic regression coefficients. Selecting the final model based on the statistical criteria for good discrimination and calibration of the predictive model. This model considered to be a good candidate, despite the decreased area under the ROC curve of the validation set. The model is “over-optimism” [38], i.e. the area under the curve will almost always be worse in the validation set than training set. The risk score is also developed for predicting risk of NVP-associated rash. This relative simple risk score, with the cutoff ≥21 points, showed a high specificity and positive predictive value. If patients had a risk score of ≥21, either EFV or PIs should be considered as an alternative in the resource limitation because of a high risk of NVP-associated rash. However, physicians should discuss risk and benefit, including close follow-up patients. High risk of unsuccessful switching from NVP to EFV was found in patients who had a history of drug allergy apart from NVP and had CD4 cell counts less than 100 cells/mm3 [39]. In the resource-limited settings, where both EFV and PIs are not available, or patients cannot tolerate both EFV and PIs, NVP might be used with caution, including slow dose escalation, closed monitoring of both clinical and liver function tests after assessing risk and benefit.
The present study had some limitations. First, there were some missing data, but it was a small proportion, and we excluded these patients from the analysis. However, we believed that model-wise deletion analysis for defining the prediction model was not bias. Second, we included only clinical characteristics and CD4 cell count in the model because these variables can apply to most of HIV-infected patients in resource-limited settings. Some laboratory variables may improve the discrimination power if they were added in the model. Third, we had to categorize continuous variables into category ones for the model building and the pre-defined cut point was arbitrary decision. This might loose information and significant association between variables and outcome. Last, this study was conducted among Thai patients, which tended to have a relative higher prevalence of NVP-associated rash. High prevalence of NVP rash in Thai patients might be explained by genetic factor. Our group recently found the strong association of HLA-B*3505 allele among Thai HIV-infected patients who developed NVP-associated rash [40]. Thus, generalization of these findings to other races should be considered cautiously.
In conclusion, a model and risk score for predicting NVP-associated rash among HIV-infected performed well in this particular study population. It might be useful for predicting the risk of rash before NVP initiation among HIV-infected patients with low CD4 cell counts especially in resource-limited setting where NVP is widely used. It is also important to monitor NVP-associated rash among HIV-infected patients with a history of drug allergy, low body weight (≤55 kg), not receiving slow dose escalation, and concomitant drug(s). In addition, further validation of this model and risk score in other populations is required.
AUTHORS’ CONTRIBUTIONS
SK carried out designed the study, developed case record form, recruited study participants, collected data, preformed statistical analysis, and drafted the manuscript. SS carried out participant recruitment and helped to draft the manuscript. KM carried out participant recruitment and helped to draft the manuscript. SW carried out participant recruitment. BS carried out participant recruitment. AC carried out study coordination. SM carried out helped to draft the manuscript. WC carried out study coordination. All authors read and approved the final manuscript.
NOTES
§Meeting: Some parts of this study were presented as a poster at the 48th Annual Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC)/ Infectious Diseases Society of America (IDSA) the 46th Annual Meeting, October 25-28, 2008, Washington D.C. [abstract H-2294].
ACKNOWLEDGEMENTS
The authors would like to thank all the attending staffs and physicians in the Division of Infectious Diseases, Department of Medicine for taking care of patients and for their supports. This work was supported by grant from Thailand Center of Excellence for Life Sciences (TCELS).


