All published articles of this journal are available on ScienceDirect.
Clinical Outcome of Lopinavir/Ritonavir (Kaletra) as HIV Second-line Treatment: A Retrospective Study at a Single Center Malaysian Tertiary Hospital
Abstract
Introduction
Virological failure remains a major challenge in treating HIV. Lopinavir/ritonavir, a protease inhibitor, is a recommended second-line option in Malaysia for patients failing first-line regimens. This study investigated the use of Lopinavir/ritonavir in terms of its efficacy in viral suppression, CD4 response, metabolic side effects, and tolerability following first-line treatment failure.
Methods
A retrospective review was conducted on adult HIV patients treated with Lopinavir/ritonavir for at least 6 months after confirmed virological failure (HIV viral load > 1000 copies/mL) on first-line therapy. Data were collected from 265 patients at Hospital Sultanah Aminah between January 2015 and December 2020, and 79 eligible patients were identified from the electronic database. Metabolic data, including lipid profile, were collected from case notes.
Results
Among 79 patients who fulfilled the criteria, most were male (n=52, 65.8%) with a mean age of 41, and the majority were of Malay ethnicity (n=51, 64.5%). Unprotected sexual intercourse was the most common transmission mode (44.3%). After one year, 96% achieved viral suppression (VL <400 copies/mL). Non-suppression was linked to non-adherence due to pill burden. Metabolic complications were common: 77% developed hypertriglyceridemia (mean triglycerides: 3.6 mmol/L), 94% had elevated LDL (mean: 3.1 mmol/L), with smaller numbers developing transaminitis (7.6%), impaired glycaemia (10.1%), hypertension (6.3%), and lipodystrophy (1.3%).
Discussion
Our study demonstrates that lopinavir/ritonavir (LPV/r) remains clinically effective at achieving viral suppression among HIV patients who have failed first-line therapy, with 76 subjects (96%) achieving viral suppression after 1 treatment. LPV/r continues to play a role in specific clinical contexts, particularly where newer antiretrovirals are not available or are contraindicated. However, LPV/r is associated with notable adverse events including GI intolerance, metabolic complications (most notably dyslipidemia), and high pill burden, all of which can negatively impact adherence and long-term treatment success.
Conclusion
LPV/r remains effective in achieving viral suppression but is associated with significant metabolic side effects. It should be reserved for individuals with limited treatment alternatives, and regimen selection should be guided by individual metabolic risk assessments. Regular metabolic monitoring and adherence support are essential, and multidisciplinary management together with further comparative studies are warranted to optimise treatment strategies that balance virological efficacy with long term metabolic safety.
1. INTRODUCTION
HIV remains an ongoing disease that causes significant morbidity and mortality. Highly active antiretroviral therapy has revolutionized the management of HIV by reducing opportunistic infection-related mortality amongst those infected and has significantly improved quality of life and survival [1]. However, virological failure remains a major challenge in treating HIV [2].
Protease Inhibitors (PI) are an effective antiretroviral drug used to treat patients who have failed first-line therapy [3]; however, the effectiveness of PI in patients who failed first-line therapy in Malaysia has not been studied. Despite the wide use of PI, it has been associated with significant side effects resulting in the discontinuation of the medication. The most common side effects are gastrointestinal, such as diarrhoea, nausea, and vomiting. As with other PIs, metabolic effects have also been reported, including the development of hypercholesterolemia, hypertriglyceridemia, increased serum glucose, and lipodystrophy [4]. The usage of PI as second-line therapy is still widely practiced.
Based on the Malaysian Consensus Guideline on Antiretroviral Therapy 2022, patients on first-line treatment undergo either routine or targeted viral load monitoring. If the viral load is detectable, the patient must be evaluated and optimised for adherence concerns. Viral load testing is repeated after 3-6 months, and a viral load > 1000 copies/ml is considered virological failure. At this stage, switching to a second line regime is recommended [5].
WHO guidelines have currently dropped boosted protease inhibitors in favour of Dolutegravir (DTG), an integrase inhibitor, for second-line therapy [6] when the DAWNING study demonstrated superiority in viral suppression, safety profile, and drug resistance outcomes [7]. However, LPV/r is still used as an alternative option in Malaysia for those who have failed the first-line regimen, in addition to other clinical scenarios.
2. OBJECTIVES
The primary objective of our study was to investigate the effectiveness of using Lopinavir/ritonavir, a PI-containing regimen, in achieving viral suppression (HIV VL < 400) among HIV-infected patients who failed first-line treatment.
The secondary objectives were:
(a) To determine the changes in metabolic profile in patients after switching to a PI-containing regimen containing compared with the previous (non-PI) regimen.
(b) To determine the tolerability or adherence of PI-containing regimen by investigating the issue of non-compliance to lopinavir/ritonavir, including intolerance to side effects.
(c) To determine the causes for non-viral suppression despite the PI regime.
3. MATERIALS AND METHODS
A retrospective study was conducted by reviewing the case notes of adult HIV patients who were treated on a PI-containing regimen with LPV/r for at least 6 months after failure of first-line treatment consisting of a combination of 2NRTIs and 1NNRTI - defined by a HIV viral load of > 1000 copies/ml of blood (WHO guidelines). A cohort of 265 patients was followed up at the Infectious Disease Clinic of Hospital Sultanah Aminah during the period of 1st Jan 2015, to December 2020. Eligible patients were identified using the electronic database registered in the pharmacy department, and metabolic data, which include lipid profile were collected from case notes. The inclusion criteria were all adult patients aged 18 or more who attended the ID Clinic and received the Hospital Sultanah Aminah PI-containing (Kaletra) regimen for at least 6 months after failure of first-line treatment; the exclusion criteria were patients on PI as first-line antiretroviral therapy (i.e., unable to tolerate Efavirenz or Nevirapine).
The following information was collected using a case report form:
- Basic demographic details (age, gender, ethnicity).
- Date of diagnosis of HIV, mode of transmission, and history of ARVs.
- HIV viral load at diagnosis of 1st line failure pre second line treatment, prior to starting PI, and 6 months post 2nd line treatment.
- CD4 level at diagnosis of 1st line failure pre second line treatment, prior to starting PI, and 6 months post 2nd line treatment.
- Metabolic profile (blood pressure, fasting serum lipid profile including LDL, TG, HDL, and TG, blood glucose, and clinical presence of lipodystrophy) at least 1 year after on lopinavir/ritonavir.
- Renal profile, specifically the level of urea and creatinine at least 1 year after lopinavir/ritonavir treatment.
- Liver function tests, specifically the level of Alanine Aminotransferase (ALT) and Aspartate Aminotransferase (AST).
- Side effects from treatment with PI.
- Reason for 1st line failure.
- Reason for non-compliant to Lopinavir/ritonavir (non-adhering to medication, defaulted follow-up, pill burden, intolerable GI side effects).
- Clinical outcome after 1 year on Lopinavir/ritonavir.
Subject identifiers were anonymized to maintain confidentiality. The collected data were analyzed using Microsoft Excel, primarily employing descriptive statistical methods. Quantitative variables were summarized using means to represent central tendencies, while categorical data were expressed as percentages to illustrate proportional distributions. Laboratory findings were tabulated and presented alongside their reference ranges to facilitate comparison and interpretation. This study is registered with the National Medical Research Register, Malaysia (NMRR ID: NMRR-21-1217-60335), and received ethical approval from the Medical Research and Ethics Committee for implementation at Hospital Sultanah Aminah.
4. RESULTS
A total of 265 subjects were screened, and 79 subjects who fulfilled the criteria were included for further analysis (Table 1). The majority of the subjects were male (n=52, 65.8%) with a mean age of 41. Most of the subjects recruited were of Malay ethnicity (n=51, 64.5%). The most common mode of HIV transmission was unprotected sexual intercourse (n= 35, 44.3%), with the majority of cases occurring through heterosexual transmission. This was followed by intravenous drug use (n=8, 10.1%) and vertical transmission (n=3, 3.8%). 76 subjects (96%) achieved viral suppression after 1 year of initiating LPV/r (defined as HIV VL < 400 copies/ml). Amongst the 3 who did not achieve viral suppression, non-adherence was reported, primarily due to pill burden. Mean viral load upon the diagnosis of treatment failure was 187,171 copies/ml (range: 910-1,067,070 copies/ml) while mean CD4 was 197 cells/mm3 (range: 5-1004 cells/mm3). The most common 1st line antiretroviral therapy prior to the initiation of LPV/r was tenofovir+ emtricitabine+ efavirenz. (Table 2). Among the cohort, metabolic complications were observed, with more than half of the subjects developing dyslipidaemia. 61 subjects (77%) recorded elevated serum triglycerides (TG > 1.7mmol/L) with a mean TG of 3.6mmol/L (range:1.72-11.8mmol/L). 74 subjects (94%) recorded elevated LDL (LDL > 3.4mmol/L), with a mean LDL of 3.1mmol/L (range: 1.9-5.8 mmol/L) (Fig. 1). A small number developed transaminitis (ALT > 55) (n=6, 7.6%), impaired glycaemia/diabetes (n=8, 10.1%), hypertension (n= 5, 6.3%), and clinical lipodystrophy (n=1, 1.3%) (Tables 3 and 4).
| Subjects (n=79) | |
|---|---|
| Mean age | 41 (range 22-68) |
| Gender | |
| Male | 52 (65.8%) |
| Female | 27 (34.2%) |
| Ethnicity | |
| Malay | 51 (64.5%) |
| Chinese | 15 (18.9%) |
| Indian | 12 (15.1%) |
| Indigenous | 1 (1.3%) |
| Mode of transmission | |
| Unprotected sexual intercourse | 35 (44.3%) |
| Intravenous drug usage | 8 (10.1%) |
| Vertical transmission | 3 (3.8%) |
| Unknown | 35 (44.3%) |
| History of 1st Line Antiretroviral Regimen | |
|---|---|
| Combivir + Efavirenz | 17 (21.5%) |
| Combivir + Nevirapine | 12 (15.2%) |
| Combivir + Raltegravir | 1 (1.3%) |
| Stavudine + Lamivudine + Efavirenz | 3 (3.8%) |
| Stavudine + Lamivudine +Nevirapine | 1 (1.3%) |
| Lamivudine+ Nevirapine+ Efavirenz | 1 (1.3%) |
| Tenofovir+ Emtricitabine+ Efavirenz | 33 (41.8%) |
| Tenofovir+ Emtricitabine+ Nevirapine | 10 (12.7%) |
| Tenofovir+ Emtricitabine+ Raltegravir | 1 (1.3%) |
| Potential reason for 1st line failure | |
| Non-compliance with medication | 32 (40.5%) |
| Social or financial-related factors | 13 (16.5%) |
| Non-compliance with clinic follow-up | 13 (16.5%) |
| Side effects of medication | 6 (7.6%) |
| Occupation related | 4 (5.1%) |
| Delayed timing of medication intake | 2 (2.5%) |
| Unknown | 9 (11.4%) |
| Mean viral load at diagnosis of failure | 187171 (range: 910, 1,067,070) |
| Mean CD4 count at diagnosis of failure | 197 (range: 5-1004) |
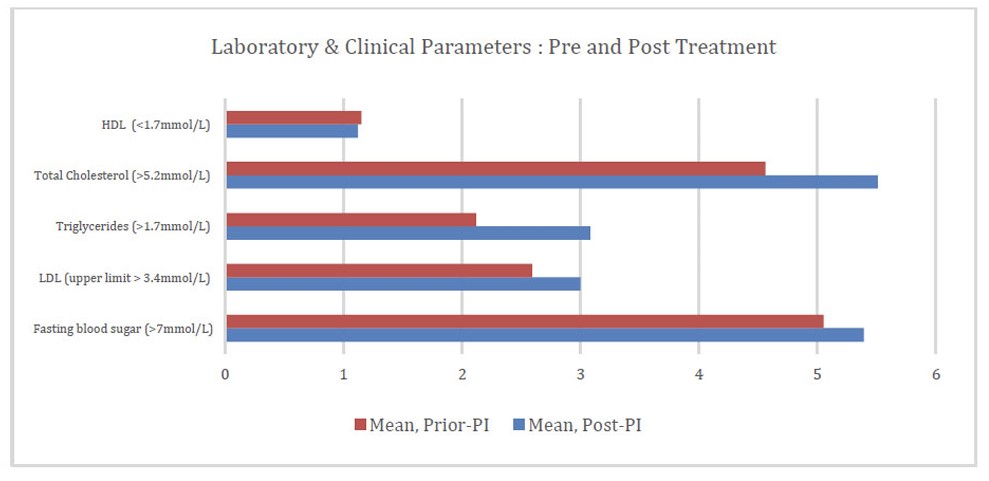
Laboratory and clinical parameters pre and post treatment.
| Compliance with Lopinavir/ritonavir | |
| Yes | 75 (94.5%) |
| No | 4 (5.1%) |
| Clinical outcomes at 6 months | |
| Viral suppression at 6 months | 72 (91.1%) |
| Mean CD4 at 6 months | 311 (range 32-1062) |
| Clinical Outcome at 1 year | |
| Viral suppressed at 1 year | 72 (91.1%) |
| Non-viral suppressed at 1 year | 7 (8.9%) |
| Other Outcomes | |
| Raised ALT (>55) | 6 (7.6%) |
| Impaired glycemia/diabetes | 8 (10.1%) |
| Hypertension | 5 (6.3%) |
| Clinical lipodystrophy | 1 (1.3%) |
| Dyslipidaemia | 74 (93.6%) |
| Parameter | Mean Prior PI | Mean Post PI |
|---|---|---|
| Fasting blood sugar (>7mmol/L) | 5.05 (n = 53, 67.1%) | 5.39 (n = 79, 100%) |
| LDL (upper limit > 3.4mmol/L*) | 2.59 (n = 49, 62.0%) | 3.00 (n = 76, 96.2%) |
| Triglycerides (>1.7mmol/L) | 2.12 (n = 49, 62.0%) | 3.08 (n = 79, 100%) |
| Total Cholesterol (>5.2mmol/L) | 4.56 (n = 50, 63.3%) | 5.51 (n = 79, 100%) |
| HDL (<1.7mmol/L) | 1.15 (n = 49, 62.0%) | 1.12 (n = 79, 100%) |
Note: *In Malaysia, there is no specific LDL-C cutoff value for the diagnosis of dyslipidaemia. Instead, current guidelines recommend that diagnosis and treatment be guided by the patient's overall cardiovascular risk profile and the presence of associated risk factors.
5. DISCUSSION
Our study demonstrates that Lopinavir/ritonavir (LPV/r) remains clinically effective at achieving viral suppression among HIV patients who have failed first-line therapy, with 76 subjects (96%) achieving viral suppression after 1 treatment. Despite its effectiveness, LPV/r has been largely replaced by newer antiretroviral agents, notably Dolutegravir (DTG), due to its less favourable side effect profile (e.g., gastrointestinal intolerance, metabolic complications), complex drug interactions, twice-daily dosing, and higher pill burden. The DAWING study highlighted DTG’s superiority over LPV/r in second-line therapy with 84% vs 70% achieving viral suppression at 48 weeks [7], leading to a shift in the WHO guidelines to recommend DTG for second-line use [6]. Nonetheless, LPV/r continues to play a role in specific clinical contexts, particularly where newer antiretrovirals are not available or are contraindicated.
The implementation of Dolutegravir (DTG) may face challenges in Low-To-Middle-Income Countries (LMICs) due to barriers such as drug supply constraints and limited access to certain formulations, including paediatric or fixed-dose combinations [6]. A global study across 35 LMICs found that while 90% of clinics had introduced DTG for first-line treatment, only 59% had done so for second-line regimens [8], emphasising that a significant proportion of patients who fail first-line therapy still lack access to DTG. Funding constraints further add to these challenges - although DTG is low-cost, LMICs may still lack the resources to replace existing ART reserves or train healthcare providers on updated treatment protocols. Given these barriers, LPV/r continues to be a practical second-line option in resource-limited settings.
Additionally, LPV/r remains an option for patients with resistance to multiple drug classes, including those who fail to respond to DTG-based regimens [6]. Whilst dolutegravir was initially anticipated to have a high genetic barrier to drug resistance [9], recent surveillance data report emerging levels of DTG resistance, with prevalence ranging from 3.9% in Uganda to 8.6% in Malawi and even reaching as high as 19.6% in Mozambique [10]. In this context, LPV/r can be utilised as a salvage option for patients with drug resistance, as multiple studies have shown that it continues to show effectiveness in treatment-experienced patients [3]. For example, a multi-centre study of 138 patients who had previously received three different antiretroviral drug classes found that LPV/r led to virological suppression in 76.3% of cases at three months and 63.2% at six months. Higher rates of virological suppression were observed in patients with less than five PI mutations (88%) compared to those with more than five (48%), suggesting that LPV/r may be even more effective when genotyping testing is available to guide treatment [11]. Furthermore, the PIVOT trial – which followed 587 patients on Protease Inhibitor (PI) monotherapy over a median period of 8 years – demonstrated the durability of PIs, including LPV/r. The study reported only one case of PI resistance (to atazanavir) [12], confirming its high genetic barrier to resistance and supporting the role of LPV/r in salvage therapy and long-term treatment.
Dolutegravir (DTG) is now the preferred option in pregnancy [6]; however, lopinavir/ritonavir (LPV/r) remains in use, particularly in cases where issues such as resistance or drug interactions limit other options. LPV/r has been shown to be effective in virological suppression during pregnancy and in preventing HIV transmission to the baby. A population-based surveillance study of 4,118 pregnant women with HIV in the United Kingdom and Ireland between 2008 and 2012 reported that 73% of women had a viral load <50 copies/ml near the time of delivery, with a Mother-To-Child Transmission (MTCT) rate of 0.5% (12/2406) [13]. When compared to other ART regimes, for example, abacavir/zidovudine/lamivudine or atazanavir, LPV/r shows similar rates of virological suppression and minimal differences in pregnancy outcomes [14, 15]. The findings support that LPV/r continues to provide a clinically useful alternative where dolutegravir is not suitable during pregnancy.
When initiating Antiretroviral Therapy (ART) in infants and children, several factors must be taken into account when selecting a regimen. These factors may include the child’s weight and age, available formulations, dosing frequency, and overall tolerability [16]. Integrase Strand Transfer Inhibitor (INSTI)-based regimens are recommended as first-line therapy, and lopinavir/ritonavir (LPV/r) is an alternative in specific clinical scenarios. LPV/r offers practical advantages, such as its availability in an oral solution and its established virological efficacy in the younger population. This was established in the IMPAACT study, which followed 451 infants and young children aged 2 months to 3 years on either an LPV/r or Nevirapine (NVP) based regimen over a 5-year period. The findings indicated that LPV/r was associated with a lower incidence of virological failure and mortality when compared to NVP. Despite concerns regarding the tolerability of LPV/r, the study reported excellent acceptability and adherence to the drug, especially in young children [17].
Beyond achieving viral suppression, managing cardiovascular disease is an increasingly important component in the long-term care of people living with HIV, particularly among those taking LPV/r, where several studies have demonstrated that LPV/r is more likely to cause metabolic disturbances compared to some other protease inhibitors, such as atazanavir or nelfinavir [18, 19]. A similar trial to ours monitored lipid parameters in HIV-infected patients on LPV/r monotherapy in resource-limited settings and found that hyperlipidaemia was common [20]. Extra precautions and regular monitoring must be considered to minimise the risk of lipid-related complications.
In 2024, the British HIV Association (BHIVA) recommended initiating statin therapy for the primary prevention of cardiovascular disease in individuals with HIV aged >40, regardless of their lipid profile or cardiovascular risk. Patients with a calculated QRISK score over 5% are prioritised for treatment [21]. The QRISK score is a UK-specific tool and may not be applicable to Malaysian populations due to differences in population characteristics and epidemiological data. Local Malaysian guidelines on CVD risk assessment in patients living with HIV are currently under review and expected to be updated in the upcoming year.
To optimise LPV/r therapy, several clinical considerations should be taken into account. Therapy initiation should be individualised according to prior antiretroviral exposure, resistance profile, and patient-specific metabolic risk factors. Once commenced on therapy, metabolic monitoring is essential – routine lipid and glucose monitoring should be integrated into routine follow-up visits to facilitate early detection and management of the metabolic complications. Finally, targeted counselling focused on adherence, lifestyle modification, and recognition of adverse effects is essential in order to support long-term treatment success.
Our study has several limitations to be acknowledged. The relatively small sample size of 79 subjects and single-centre design limit the extent to which these findings can be generalised to the broader population of individuals living with HIV, particularly those living in different geographic or ethnic settings. Due to the retrospective nature of the study, our study is subject to incomplete or missing data, which may compromise the accuracy and reliability of outcome assessments. Additionally, the lack of a control arm prevents direct comparison with alternative antiretroviral regimens, thereby limiting the ability to determine whether the observed metabolic complications or virological outcomes are uniquely attributable to LPV/r. These limitations suggest that while our study provides valuable insights into the use of lopinavir/ritonavir, the findings should be interpreted with caution, given the methodological constraints.
CONCLUSION
In summary, lopinavir/ritonavir continues to demonstrate effectiveness in achieving viral suppression in patients with first-line treatment failure. Given its strong genetic barrier to resistance and established efficacy across different patient populations, LPV/r remains a clinically valuable option in the context of resource-limited settings with specific patient populations, including pregnant and young patients. It is also a practical fallback in cases where dolutegravir availability is limited or in cases of emerging resistance. Its use should be reserved for individuals with limited treatment alternatives, and regimen selection should be guided by individual metabolic risk assessments and profiles.
However, LPV/r is associated with notable GI intolerance, metabolic complications (most notably dyslipidemia), and high pill burden, which may negatively impact adherence and long-term treatment success. Multidisciplinary team management, including adherence support, appropriate counselling, and timely cardiovascular monitoring, is recommended as standard of care.
AUTHORS’ CONTRIBUTIONS
The authors confirm their contributions to the paper as follows: C.K.: Performed data analysis and drafted the manuscript; L.S.: Served as the project lead investigator and was responsible for recruitment, data analysis, and drafting of the manuscript; M.Z.: Provided supervision and contributed to drafting the manuscript; E.O.: Responsible for the study conception, supervision, and final correction of the manuscript. All authors reviewed the results and approved the final version of the manuscript.
LIST OF ABBREVIATIONS
| HIV | = Human immunodeficiency virus |
| ART | = Antiretroviral therapy |
| ARVs | = Antiretrovirals |
| PI | = Protease inhibitor |
| LPV/r | = Lopinavir/ritonavir |
| DTG | = Dolutegravir |
| NRTI | = Nucleoside reverse transcriptase inhibitor |
| NNRTI | = Non-nucleoside reverse transcriptase inhibitor |
| VL | = Viral load |
| LMIC | = Low-and-middle income countries |
| WHO | = World Health Organisation |
| CVD | = Cardiovascular disease |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study is registered with the National Medical Research Register, Malaysia (NMRR ID: NMRR-21-1217-60335), and received ethical approval [KKM/NIHSEC/ P21-1417 (4)] from the Medical Research and Ethics Committee for implementation at Hospital Sultanah Aminah.
HUMAN AND ANIMAL RIGHTS
All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1964, as revised in 2024.
CONSENT FOR PUBLICATION
Patient consent was waived by both Ethical Committees due to the use of de-identified data, ensuring that no patient identifiers were included in the study.
AVAILABILITY OF DATA AND MATERIALS
The data of current study are available from corresponding author, [E.O], on a reasonable request.
ACKNOWLEDGEMENTS
The authors would like to thank the Director General of the Ministry of Health Malaysia for granting them permission to undertake and publish this study. They also extend their thanks to the HSAJB staff for their valuable support in providing access to patient records throughout the study.


