All published articles of this journal are available on ScienceDirect.
Important Risk Factors of Liver Cirrhosis in HIV and Hepatitis C Coinfected Patients: A Systematic Review
Abstract
Introduction
Hepatitis C virus (HCV) is the leading cause of chronic hepatitis and liver fibrosis. Due to shared modes of transmission with human immunodeficiency virus (HIV), HIV-HCV coinfection is also common worldwide. Multiple studies have shown that the rates of liver fibrosis and associated complications increase considerably in this sub-population compared to a single HCV infection. Thus, in this study, we aimed to conduct a systematic review of possible associated important risk factors of accelerated liver cirrhosis among HIV-HCV coinfected subjects.
Methods
A systematic review of published studies relevant to the main risk factors of liver cirrhosis progression in HIV and hepatitis C coinfected patients was performed using databases of PubMed, Web of Science, Scopus, and Embase were searched using keywords and their combinations. We retrieved all the relevant papers and reports published in English till 27 June 2022, which were examined by applying inclusion/exclusion criteria for data extraction after a two-step screening process.
Results
The long-term or chronic hepatitis C and HIV coinfection is a substantial risk factor for Cirrhosis. Primary etiologies identified causing fibrosis, and the rapid progression of Cirrhosis in HIV/HCV coinfected patients include high-risk alcohol consumption, chronic elevation of ALT, AST, Aspartate Aminotransferase to Platelet Ratio Index (APRI) and Gamma-glutamyl Transferase (GGT), Body Mass Index (BMI), older age, high HIV and HCV viral loads, lower CD4+ count (<250/mm3), and male gender. Comorbidities such as diabetes, hypertension, hyperlipidemia, and high visceral fat area are suggested etiologies of cirrhosis.
Conclusion
The results showed that HIV accelerates the progression of HCV-related liver disease independent of its effect on the immune system. This effect is somehow dependent on age, gender, BMI, duration of HIV infection, and CD4 count.
1. INTRODUCTION
Hepatitis C virus (HCV) is a common cause of acute and chronic hepatitis and a leading cause of liver cirrhosis, and hepatocellular carcinoma (HCC) worldwide. The global HCV infection prevalence has been estimated at 2.5%, infecting 177.5 million people [1, 2]. According to the World Health Organization (WHO), 58 million people live with chronic HCV infection globally, and approximately 1.5 million people get HCV every year. HCV is a bloodborne virus, and its main route of infection is blood exposure from unsafe injection, unscreened transfusions, IV drug abuse, and sex [3]. Moreover, although remarkable advances have been achieved in HCV treatment, and it is considered a curable infectious disease with a viral eradication rate of more than 95% among HCV-infected cases, it remains a global health burden that claimed the lives of approximately 290 000 people in 2019 [4].
On the other hand, due to shared transmission routes, HCV and human immunodeficiency virus (HIV) coinfection is common, and according to the latest studies, the HIV-HCV coinfection rate among HIV-infected individuals of the general population samples is 2.4%. At the same time, it reaches a peak of 82.4% among HIV-infected IV-drug users. Also, different studies have estimated that between 2.2 to 7 million individuals are co-infected with HIV and HCV globally [5-7]. In addition, the odds of HCV infection are six-fold higher among HIV-infected subjects than that of HIV-negative people [5, 8]. Moreover, according to WHO, 70% of HCV-infected patients develop chronic HCV infection,e. Among chronic HCV-infected cases, the 20-year cirrhosis risk ranges between 15% to 30%. At the same time, it has been shown that HIV-HCV coinfection changes the natural course of HCV infection in a way that its chronicity rate soars to over 90% among HIV patients [9, 10].
Consequently, after this increase in chronicity, HCV disease progression occurs much faster and causes higher mortality rates, histological fibrosis/cirrhosis, and decompensated liver disease among HIV-HCV coinfected patients compared to single HCV-infection [11, 12]. One meta-analysis reported a significant elevation in the Relative Risk (RR) of liver disease. It measured a combined RR of 6.14 and 2.07 for decompensated liver disease and liver cirrhosis, among HIV-HCV coinfected patients, respectively [11, 13]. Furthermore, it has been shown that in addition to liver-associated deaths, all-cause mortalities were also increased among HIV-HCV coinfected subjects compared to either HIV or HCV infection alone [14, 15].
Many studies have focused on cellular and molecular mechanisms, and indicators of accelerated liver fibrosis among HIV-HCV coinfected patients [16-22]. One study reported that HIV upregulates HCV replication, augments HCV-related TGF-β1 release from hepatocytes, independently generates reactive oxygen species (ROS), and induces hepatocyte apoptosis (in combination with HCV) [16]. In addition, in multiple studies an extensive variety of potential risk factors have been associated with progressive cirrhosis progression in HIV-HCV coinfected patients as follows; long-term duration of coinfection, high HIV and/or HCV viral load, lower CD4+ and/or CD8+ T-cell count, high body mass index (BMI), older age, high-risk alcohol consumption, male gender, and IV drug injection [17-22]. While to the best of our knowledge, no systematic review is available on the risk factors of liver cirrhosis among coinfected individuals. Therefore, in this study, we aimed to conduct a systematic review of the current literature to shed light on the associated important risk factors of HIV-HCV coinfection on liver cirrhosis.
2. MATERIALS AND METHODS
In this study, we intend to determine the important risk factors of liver cirrhosis in HIV and hepatitis C coinfected patients. To achieve this goal, we have reviewed studies published in English from inception until 27 June 2022. For the purpose of reliability and validity, we have adhered our study to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist.
2.1. Data Sources
All the relevant papers and reports published in English until 27 June 2022 were used in a systematic search using keywords in the online databases such as PubMed, Web of Science, Scopus and Embase. Our search strategy employed multiple combinations of keywords, as follows:
• (“HIV”(mesh) OR “Acquired Immunodeficiency Syndrome”(mesh) OR Acquired Immune Deficiency Syndrome Virus(tiab) OR Acquired Immunologic Deficiency Syndrome*(tiab) OR Acquired Immune Deficiency Syndrome*(tiab) OR Acquired Immuno-Deficiency Syndrome*(tiab) OR Acquired Immuno- deficiency(tiab) OR AIDS(tiab) OR HIV(tiab) OR HTLV-III(tiab) OR Human T Lymphotropic Virus Type III(tiab) OR Human T-Lymphotropic Virus Type III(tiab) OR Human Immunodeficiency Virus*(tiab) OR Human Immuno deficiency Virus*(tiab) OR Human T Cell Lymphotropic Virus Type III(tiab) OR Human T-Cell Leukemia Virus Type III(tiab) OR LAV-HTLV-III(tiab) OR Lymphadenopathy-Associated Virus*(tiab) OR Lymphadenopathy Associated Virus(tiab) OR Lymphadenopathy Associated Retrovirus (tiab) OR LAV(tiab) OR LAV (AIDS)(tiab) OR Immuno- deficiency associated virus(tiab))
• (“Hepatitis C” (mesh) OR “Hepatitis C, Chronic” (mesh) OR Hepatitis C(tiab) OR Parenterally-Transmitted Non-A Non-B Hepatitis(tiab) OR Parenterally Transmitted Non A Non B Hepatitis(tiab) OR PT-NANBH(tiab)) AND (“Liver Cirrhosis” (mesh) OR Cirrhosis(tiab))
• (A) and (B)
Also, We performed a manual search of the references mentioned in the selected studies and review articles to notice extra relevant publications.
2.2. Study Selection
We included original English articles writing about the long-term coinfection of HIV and Hepatitis C as the strongest factor for the development of liver cirrhosis. Also, other factors such as age, gender, alcohol consumption and viral load were investigated in these articles. We did not include case reports, case series, review articles, and comments. After removing duplicated ones, in order to improve our screening process, we implemented two independent steps. The first one was to screen and preliminarily contain relevant articles with regard to their titles and abstracts. This step was accomplished by four researchers. In the second and more insightful step, researchers went through the screening of full texts of the articles, which met the first step and fulfilled the eligibility criteria for application. The relevant papers went through data extraction. The items of inclusion/exclusion criteria which have been determined for the study are as follows:
The inclusion criteria:
• To be original
• Human study only
• To be published in the English language
• To be peer-reviewed prior to publication
The exclusion criteria:
• Papers deficient of full texts like abstract papers and conference abstracts
• Ongoing trials and studies without yet published data
• Duplicated literature
2.3. Data Extraction
ID, the first author (reference), Type of study, Country, Study Population, Gender, Age, Cd4 count, Viral load (HIV, HCV), Inflammatory cytokines, Alcohol consumption, Genetic factors, Genetic factors, HIV/HCV coinfection Duration, Visceral abdominal fat and other were recorded in Tables 1 and 2, which were collected by other five independent researchers and subsequently organized in tables, which were the most significant risk factors in liver cirrhosis. All authors cross-checked the selected articles to avoid any duplications or overlaps in the content.
2.4. Quality and Risk of Bias Assessment
As stated earlier, this study adhered to the PRISMA checklist to ensure the quality and accuracy of selected papers and outcomes. Two independent researchers examined the quality of the articles and the probable risk of bias. The full text of selected articles was read, and the low-quality articles were not imported, and analytically, 37 articles were of good quality (79.4%), and the other 10 articles were of fair (20.6%) quality.
3. RESULTS
3.1. Selection of Studies
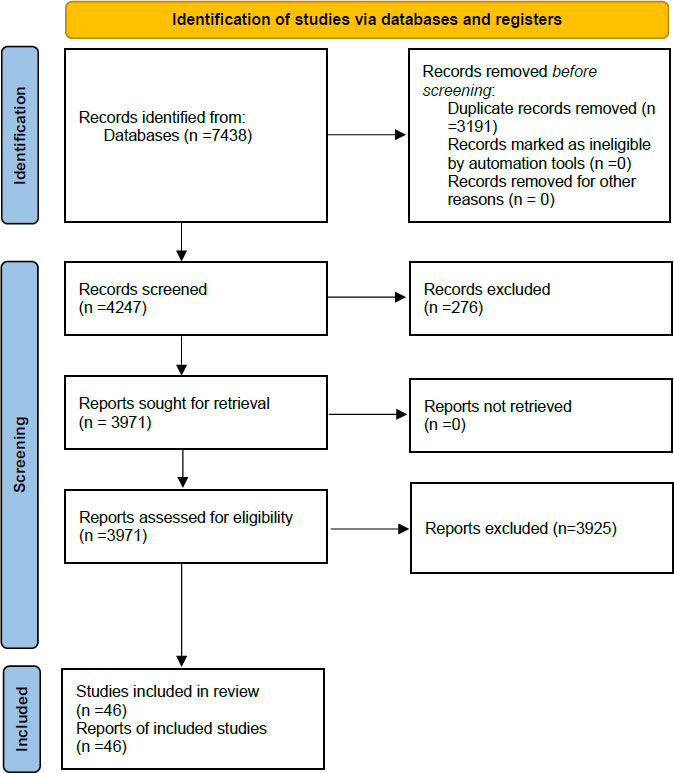
A total of 7,438 studies were identified after the primary search, of which 3,191 were removed due to duplication. The full texts of 276 papers were not retrievable. Hence they were excluded. Titles and abstracts of 3,971 papers were then checked for their eligibility for this study and 3,908 articles were excluded Irrelevant by Title (683), Irrelevant by Abstract (299), Papers published before the year 2012 (1,907), Review articles about Cirrhosis in HIV/HCV patients (196), Conference abstracts (757), Case reports [66]. Full texts of 63 published articles about Cirrhosis in HIV/HCV patients were studied, which resulted in the exclusion of 16 more articles due to irrelevance to our study. The PRISMA flow diagram in Fig. (1) illustrates this process. The qualitative systematic review was performed on 46 articles.
| ID | The First Author (Reference) | Type of Study | Study Population |
|---|---|---|---|
| 1 | Oliver [41] | Retrospective cohort study | 5985 HIV/HCV co-infected veterans |
| 2 | Pérez-Is [49] | Prospective cohort study | 160 HCV-monoinfected, 214 HCV/HIV coinfected, Total: 374 |
| 3 | Polo [48] | Case-Control Study | 34 HIV/HCV co-infected patients, 16 with minimal fibrosis (F0/F1], 18 with advanced fibrosis (F4], 20 Healthy volunteers |
| 4 | Puoti [36] | Secondary data | 4,059 patients |
| 5 | Quaranta [33] | Prospective cohort study | 244 HIV/HCV coinfected patients (93 with cirrhosis), 2870 HCV monoinfected patients |
| 6 | Re III [50] | retrospective cohort study | 10359 patients, 4280 HIV/HCV patients, 6079 HCV-monoinfected patients |
| 7 | Fernandez-Montero [17] | retrospective cohort | 545 HIV–HCV-coinfected, 99 with LFP, 446 without LFP |
| 8 | Fernandez-Montero [18] | Retrospective cohort | 1147 HIV infected, 521 patients (45.4%) were HCV antibody positive, 85, (7.4%) were HBsAg positive, and another 17 (1.5%) were antiHDV positive |
| 9 | Fernandez-Montero [29] | Observational, retrospective | 545 hiv/HCV coinfected |
| 10 | Fernandez-Rodriguez [27] | Retrospective follow-up cohort study. | 190 HIV/HCV-coinfected, 25 did not develop fibrosis (F0) (non-progressor), 165 patients developed fibrosis(F≥1] (progressors) |
| 11 | Ferreira [34] | Cross sectional | 354 HIV-infected, 93 monoinfected, 261 coinfected: 40.1% HIV/HBV, 19.5% HIV/HCV and 14.1% HIV/HBV/HCV |
| 12 | Feuth [19] | Cross-sectional | 89 subjects, including 18 chronic HCV monoinfected patients, 10 HIV-1 monoinfected patients, 14 HIV/ HCV coinfected patients, 18 HBV infected patients, 14 PBC patients and 15 healthy controls |
| 13 | Foca [30] | Cohort | 1433 HIV/HCV coinfected |
| 14 | Franco [20] | Cross-sectional | 46 HIV-1/HCV coinfections, 20 Non progressing, 26 progressing in mean of 10.3 years And 21 uninfected |
| 15 | Medrano [51] | cross-sectional study | 220 HIV/HCV-coinfected |
| 16 | Medrano [52] | cross-sectional study | 238 HIV/HCV-coinfected patients, 32 healthy controls, 39 HIV-monoinfected |
| 17 | Merchante [53] | prospective cohort | 239 HIV/HCV-coinfected |
| 18 | Molina-Carrión [28] | prospective cohort | 44 HIV/HCV-coinfected, 9 HCV-monoinfected |
| 19 | Moqueet [54] | prospective cohort | 485 HIV/HCV-coinfected |
| 20 | Moqueet [45] | case-cohort study | 679 HIV-HCV coinfected |
| 21 | Nguyen Truong [37] | cross-sectional study | 104 HIV-HCV coinfected |
| 22 | Nunez-Torres [44] | cross-sectional and longitudinal | 337 HIV-HCV coinfected |
| 23 | Márquez-Coello [55] | Cohort study | HCV monoinfected:20, HCV/HIV co-infected:66, Control:15 |
| 24 | Matas [56] | Cohort study | 63 |
| 25 | Giovanni Mazzola [57] | observational, retrospective study | HCV mono-infected (n=1937], HIV/HCV co-infected (n=238] |
| 26 | José A. Mira [58] | Prospective cohort | 166 HIV-HCV coinfected, 43 SVR, 123 non-SVR |
| 27 | Marco Merli [59] | n observational retrospective | 646 coinfected, LS<13kpa n=474, LS>=13kpa n=172 |
| 28 | French [60] | Cross-sectional | HIV/HCV coinfected patients (n=44] |
| 29 | French [61] | Prospective cohort | HIV/HCV coinfected patients (n=126] |
| 30 | Garcia-Broncano [40] | Cross-Sectional | 238 HIV/HCV co-infected patients, 39 HIV monoinfected patients, 32 healthy donors negative for HIV, HCV and HBV |
| 31 | Frias [46] | Retrospective and longitudinal cohort | HIV/HCV coinfected (n=104] |
| 32 | Fuster [47] | Cross-Sectional | 308 |
| 33 | Gad [35] | Cross-Sectional | HIV-monoinfected (n=169], HIV/HBV coinfected (n=20], HIV/HCV coinfected (n=39], HIV/HBV/HCV, Triplet infection (n=13] |
| 34 | Mandorfer, Mattias [62] | 86 HIV/HCV | |
| 35 | Shmagel [39] | Case-control Study | [1] HIV/HCV coinfected patients (n = 42]; |
| 36 | Soldevila [42] | Cross-Sectional | 115 HIV/HCV-coinfected patients |
| 37 | Steininger [21] | Retrospective Cohort | 178 MSM |
| 38 | Tabernilla [43] | Clinical Cohort | 140 HCV monoinfected, 212 HIV/HCV-coinfected patients, Total = 352 |
| 39 | Valcour [32] | Cross-Sectional | 770 (52%) only HIV infected,, 73 (5%) only hepatitis C virus, (HCV) infected, 235 (16%) HIV/HCV coinfected, and 401 (27%), Uninfected |
| 40 | van Santen [24] | Clinical Cohort | 173 study participants |
| 41 | van Santen [26] | Clinical Cohort | Study population I 85, Study population II 55, Total = 140 PWUD |
| 42 | Vermehren [23] | Cross-Sectional | 202 consecutive HIV-infected individuals |
| 43 | Vogel [38] | Prospective Cohort | 38 participants |
| 44 | Wei [22] | Retrospective Cohort | 955 HIV-infected patients (808 (84.6%) were HIV-monoinfection) (125 (13.1%) were HIV/hepatitis B virus (HBV) coinfection) (29 (3.0%) were HIV/hepatitis C virus (HCV) coinfection) |
| 45 | Yaya [31] | Prospective Cohort | 1,019 HIV/HCV co-infected patients |
| 46 | Yuh [25] | Clinical Cohort (Thesis) | 93 HIV/HCV co-infected patients |
| ID |
Gender (n, percent) |
Age (Mean±SD) |
CD4 Count | Viral Load | Inflammatory Cytokines | Alcohol Consumption | Genetic Factors | HIV/HCV Co-infection Duration | Visceral Abdominal fat | HCC Developement | Effect of SVR | ||
| Male | Female | HIV | HCV | ||||||||||
| 1 | 5985 (100%) |
- | 45 | <350 cells/μL (48.9%) Patients with CD4 count <200 cells/μL and ALT ≤40 IU/L or >40 IU/L had greatest risk of cirrhosis. |
Suppressed HIV viral load (more than 80% of the time) In 1079 (29.0%) patients without cirrhosis and 449 (19.8%) cirrhotic patients |
N/A | Significant association in patients with ALT >40 IU/L |
Diabetes (16%) Hypertension and low-HDL (> 50%) |
N/A | BMI >30 853 (22.9%) without cirrhosis 450 (19.9%) with cirrhosis |
no statistical difference between the two groups | No statistical difference between two groups in HCV treatment in none, no SVR and SVR groups | |
| 2 | 264 (70.6%) | 110 (29.4%) | 50.81 (50.02–51.59) | 626.60 cells/µL (95% CI 575.89–677.30) | HIV: 0.677 (0.476–0.878) log copies/mL HCV: 5.908 (5.713–6.102) Log copies/mL |
MMP-2 (-1306 C/T) variant TT genotype |
N/A | HCV genotype 1 124 (72.1%) |
N/A | N/A | N/A | N/A | |
| 3 | F4: 14 (78%) |
F4: 4 (22%) F4: 4 (22%) |
F4: 51.2 (35 to 64) |
F4: 563.8 (54 to 1278) cells/μL | HCV viral load: 6.12 (4.74 to 7.38) log10 copies |
Augmentation of PD-1 | N/A | N/A | For HCV: 16.3 (3 to 31) years For HIV: 19.3 (3 to 30) years |
N/A | N/A | none of them achieve sustained virologic response | |
| 4 | N/A | N/A | Lower risk of seroconversion in older age | No association with CD4 counts | No association with HIV viral load | N/A | Alcohol intake (RH 20.79 95%CI 1.57-274.80) | N/A | N/A | N/A | N/A | N/A | |
| 5 | 182 (74.6%) | 62 (25.4%) | 52 (32–77) | N/A | N/A | N/A | Current73 (33.0 %) Past 44 (19.9%) |
Diabetes 26 (10.7%) |
N/A | Overweight 58 (23.8%) Obese 11 (4.5%) |
Age, male sex, lower albumin levels, genotype 3 and serum anti-HBc positivity were independently associated with HCC incidence | Similar rates of SVR12 were observed in coinfected (94.9%) and monoinfected (94.8%) patients with liver cirrhosis | |
| 6 | 4214 (98.5%) | 66 (1.5%) |
48 (44–52) | <0.200 109 cells/L | 3186 (74.5%) with HCV RNA level: 400 000 IU/mL and/or 1 000 000 copies/mL Higher rates of decompensation for HIV RNA levels: <1000 copies/mL or <400 copies/mL |
N/A | 1130 (26.4) | Baseline Diabetes 318 (7.4) |
N/A | N/A | N/A | N/A | |
| 7 | 71.5% 20.3% of patients. with LFP 13% of patients. without LFP |
28.5% | 41.5±5.3 41.3±5.2 in patients with LFP 41.6±5.3 in patients without LFP |
519 ±284 cells/µl 470±264 in patients with LFP 530±288 in patients without LFP |
HIV RNA: 2.34±0.93 in patients with LFP 2.21±1 in patients without LFP HCV RNA: 5.6±1.86 in patients with LFP 4.7±2.23 in patients without LFP |
ALT: 82.3±49.8 in patients with LFP 101.4±30.8 in patients without LFP |
8.4% 32.6% of patients. with LFP 15.5% of patients. without LFP |
N/A | N/A | N/A | N/A | N/A | |
| 8 | 80.6% | - | 42.1±7.1 | 566 ±310 cells/µL | - | ALT: 53.6±56.2 |
6.9% | N/A | N/A | N/A | patients who achieved HCV clearance, exhibited event-free survival rates that did not differ from those in HCV-monoinfected controls. | SVR following hepatitis C therapy in HIV/HCV-coinfected patients was protective (p-value = 0.03) | |
| 9 | 71.5% | 28.5% | 41.5±5.3 | 519 ± 284 cells/ml | Undetectale plasma HIV-RNA (%) 88 Plasma HCV-RNA (log IU/ml) 4.9 2.2 | Mean ALT was 65±54 IU/ml | 8.4% | N/A | N/A | N/A | N/A | During follow-up, 335 patients (63%) were treated with PegIFN and RBV and 132 (39.4%) of them achieved SVR which was significantly associated with the development of liver-related events | |
| 10 | F=0: 16 F≥1 126 |
F=0: 9 F≥1 39 |
F=0: 43.2 F≥1: 39.6 |
F=0: 153 F≥1 185 |
N/A | N/A | 48% of F=0 58.7% of F≥ |
N/A | 25 patients with a median of 25 years (Non-progressors) 165 with a median of 21.3 year (progressors) |
N/A | N/A | N/A | |
| 11 | 56.5% | - | Median: 46 92% >40 years |
Median 514 |
HIV viral load undetectable: 58/61 <50copies/ml |
AST median: 45 ALT median: 50 |
N/A | N/A | N/A | N/A | N/A | N/A | |
| 12 | 86% | Median 48 | HCV- RNA median: 4.5e5 IU/ml | ALT median 65 | CD4+ CD8+ |
N/A | N/A | N/A | N/A | N/A | |||
| 13 | 74.39% | 25.61% | Median: 34 | Median: 292 | HIV-RNA log10 (median) 4.06 HCV-RNA log10 (median) 5.84 |
Higher CD4+ T-cell protected from clinical events γGT is a negative predictor for any outcome |
N/A | N/A | N/A | N/A | N/A | N/A | |
| 14 | Non-progressing: 70% Progressing 73% |
- | Non-progressing: 39 Progressing: 36 |
Non-progressing: 624 Progressing: 481 |
HCV: Non-progressing: 5.6 Progressing: 5.7 |
AlT : Non-progressing: 51 Progressing: 84 |
N/A | patients with coinfections displayed 16 upregulated and 1 downregulated miRNAs | N/A | N/A | N/A | N/A | |
| 15 | 162 (73.6%) | - | 162 (73.6%) | 467 (324; 672) | N/A | N/A | N/A | ADAR1 gene | N/A | N/A | N//A | N//A | |
| 16 | 187 (87.6%) | - | 49(46,52) | 547 (394,803) | HIV-RNa>50:30(12.6%) HCV-RNA>500000 :191 (80.2%) |
T-cell activation (CD4+CD38+ and CD8+CD38+), bacterial translocation (sCD14), inflammation (IL-1b, IL-6, IL-8, IL-18, IP-10), endothelial dysfunction (sVCAM1, sICAM1, and sTNFR1), and coagulopathy (PAI-1)] than healthy controls and HIV-monoinfected patients | N/A | N/A | N/A | N/A | N/A | N/A | |
| 17 | 215 (90%) | - | 44 (41-48) | CD4 cells/mL: 406 (247-615) | HCV RNA load (log10 IU/mL): 6.16 (5.68-6.67) HIV RNA load < 50 copies/mL, no. (%): 166 (70) |
- | Daily alcohol intake > 50 g/day, no. (%) 30 (12) | N/A | N/A | N/A | 8% of patients with a baseline LS below 40 kPa developed a decompensation and/or HCC during follow-up, the respective figure for those individuals with a baseline LS above or equal to 40 kPa was 29% | SVR was achieved in 19 (23%) Liver-related mortality and/or transplantation tended to be lower in those patients who achieved SVR during follow-up |
|
| 18 | HIV/HCV-coinfected: 49 (81.7%) HCV-monoinfected: 17 (56.7%) |
- | HIV/HCV-coinfected: 51.7 (48.7–53.8) HCV-monoinfected: 58.5 (52.3–69.6) |
439 (234–717) | Log10 HCV RNA (IU/mL) HIV/HCV-coinfected: 6.2 HCV-monoinfected: (5.7–6.7) 6.11 (5.50–6.41) |
N/A | Alcohol drinker (>50 g/day) HIV/HCV-coinfected :37 (61.7%) HCV-monoinfected: 9 (30%) |
RTL was significantly lower in HIV/HCV-coinfected patients | N/A | N/A | N/A | The relative telomere length(RTL) was significantly lower in HIV/HCV-coinfected than in HCV-monoinfected patients after SVR | |
| 19 | 335 (69%) | - | 44 (38–49) | 381 (260–550) | N/A | N/A | Current alcohol drinker:234 (48) | IFN-λ genotype: rs12979860CC: 202 (42) rs8099917TT: 297 (61) rs8103142TT: 214 (44) |
N/A | N/A | N/A | N/A | |
| 20 | - | 187 (28) | 44 (39–49) | 400 (270–568) | Undetectable HIV viral load, (<50 copies/ml) : 395 (59) | N/A | Current alcohol drinker:333 (49) | IFNL genotype rs8099917 TT: 333 (60) | N/A | N/A | N/A | N/A | |
| 21 | 99 (95.2%) | - | 35.8 (32.7–39.6) | 504 (361–624) | Undetectable HIV-RNA (n, %): 98 (94.2) Detectable HCV-RNA, n (%): 93 (89.4) |
N/A | 70 (67.3) | N/A | N/A | N/A | N/A | N/A | |
| 22 | 287 (86.4%) | - | 47 (43–50) | 479 (311–710) | Serum HIV-RNA <50 Copies/mL, n (%): 261 (78.6) HCV viral load: 6.2 (5.6–6.6) |
- | Alcohol intake ≥ 50g/day, n (%): 59 (18.4) | rs738491 (SAMM50) rs12743824 (LPPR4) rs738409 (PNPLA3) |
N/A | N/A | N/A | 37.5% of individuals previously received treatment against HCV infection without SVR | |
| 23 | HCV monoinfected:8(35) HCV/HIV co-infected:58(88) Control:10(67) |
- | HCV monoinfected:60 HCV/HIV co-infected:54 Control:55 MEDIAN |
494/ml (312–791) | HCV VIRAL LOAD: monoinfected:5.6 HCV/HIV co-infected:6.1 100% had undetectable HIV viral load (< 50 copies/ml) |
N/A | N/A | N/A | N/A | N/A | N/A | N/A | |
| 24 | 41(67.2%) | 20(32.8%) | N/A | N/A | Low:20(36.3) High:35(63.7) |
N/A | 3(5.2) | N/A | N/A | N/A | N/A | AST and ALT levels and the frequency of HCV genotype 3 infection (all with a P < 0.05) were higher in patients with a SVR Patients with the CC genotype had higher SVR rates than those with the CT or TT genotypes (P = 0.008) |
|
| 25 | HCV mono-infected: 1352 (69.8%) HIV/HCV co-infected: 183 (76.9%) |
- | HCV mono-infected:51 HIV/HCV co-infected:52 |
N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | |
| 26 | SVR: 37 (86) Non SVR: 105 (85) |
- | SVR: 42 NON SVR: 43 |
SVR: 477 NON SVR: 418 |
SVR: HCV RNA log IU/ml: 5.5 Undetectable HIV load: 41 (95%) NON SVR: HCV RNA log IU/m; 6 Undetectable HIV load: 105 (85%) |
N/A | N/A | N/A | N/A | N/A | the frequency of cases of HCC in subjects who reached SVR was lower than in cirrhotic patients without this response, there were no statistically significant differences between both groups. | Two of 43 patients included in the SVR group developed liver decompensation, specifically 1 episode of ascites and 1 case of HCC, as compared with 33 of 123 individuals without SVR (P = .002) | |
| 27 | LS<13kpa: 329 (69%) LS>=13kpa: 134(78%) |
- | LS<13kpa: 48 LS>=13kpa: 49 |
LS<13kpa: 552 (27%) LS>=13kpa: 448 (27%) |
LS<13kpa: HIV RNA<50 copies/ml: 368 (82%) HCV RNA log IU/ml: 5.91 LS>=13kpa: HIV RNA<50 copies/ml: 132 (85%) HCV RNA log IU/ml: 5.79 |
LS<13kpa: ALT: 57 LS>=13kpa: ALT: 82 |
N/A | N/A | N/A | N/A | N/A | N/A | |
| 28 | 0 (0%) | 44 (100%) |
Progressors(n=21) 42.4±7.4 Non-progressors(n=23) 40.8±5.7 |
Progressors (421 cells/mm3) Non-progressors (526 cells/mm3) |
N/A | Progressor had significantly higher sCD14, IL-6 and I-FABP levels | Progressors yes (n=14) Non-progressor Yes (n=19) |
N/A | 5 years | N/A | N/A | N/A | |
| 29 | 0 (0%) | 126 (100%) |
56.3 years | 636.5 (331.6) | Log HIV RNA (copies/mL), mean (SD) 5.39 (2.1) Log baseline HCV RNA (IU/mL), mean (SD) 14.15 (1.7) |
N/A | N/A | N/A | N/A | N/A | N/A | N/A | |
| 30 | 83(81.4%) | 47 | 49 (48-50) |
>500 cells/mm3 = (n=32 HIV monoinfected ) | N/A | Higher values of plasma cytokines levels and IL-10, IFN-y, memory Tregs and CD4+ Tregs. | N/A | N/A | N/A | N/A | N/A | N/A | |
| 31 | 89 (85.6%) |
15 (14.4%) |
48 (44-53) |
<350 cell ml−1 = (n=43.9%) >350 cell ml-1 = (n=42.9%) |
N/A | N/A | 0-20g/d = (45.2%) 20-50g/d = (50%) ≥50g/d = (26.7%) |
N/A | N/A | N/A | N/A | N/A | |
| 32 | 73% | 27% | 42±7.3 years | <200 (18.7%) 200-500 (41.9) >500 (39.4%) |
HIV RNA <500 (44.2%) ≥500 (55.8%) HCV RNA (48.9%) |
Higher levels of IL-6 | Heavy Alcohol use 61(32.9%) | Diabetes mellitus (5.8%) | N/A | N/A | N/A | N/A | |
| 33 | 97 (40.2%) |
144 (59.8%) |
38.19±8.5 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | |
| 34 | - | - | - | CD4nadir <200 cells × μl−1 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | |
| 35 | (1) HIV/HCV coinfected patients (61.9%) | (1) HIV/HCV coinfected patients (38.1%) | 32 ±2 years | (1) HIV/HCV coinfected patients = 350 (260–450) cells/µL; | (1) HIV/HCV coinfected patients [HIV < 50] copies/mL, [HCV= 6.21] log10 copies/mL |
Plasma levels of the inflammatory cytokines IL-6 and IP-10, the monocyte/macrophage markers neopterin and sCD163, and sTNF-RII were higher in HIV/HCV coinfected patients | N/A | N/A | 11 years | N/A | N/A | N/A | |
| 36 | 73% | 27% | 50 (47–54) years | 566 (390–772) cells/ µL | Detectable HIV Viral Load, N (%) = 13 (11) HCV= 6.28 (5.71–6.76) log10IU/mL |
N/A | Yes 9 (8%) |
N/A | N/A | VFA, median (IQR) = 167.5 (124–239) | N/A | N/A | |
| 37 | 100% | 0 (0%) | 39 (32-45) years | 514 (386-676) (/mm3 ) |
HIV RNA viral load (log10 copies/mL) = 4.44 (3.15-5.06) HCV RNA viral load = 6.09 log10 IU/mL |
elevated ALT levels | Yes 22 (12.9%) |
N/A | >11 years | N/A | No patient developed liver decompensation or HCC, had a liver transplant, or died of a liver-related cause of death during the observation period. |
Incident rates for significant fibrosis or cirrhosis were higher for patients without SVR (IR=16.5/100 person-years) compared to those with spontaneous clearance (IR=7.0/100 personyears, P=.17), or SVR (IR=6.7/100 person-years, P=.007) | |
| 38 | 73.6% | 26.4% | 49.95± 9.15 years | N/A | N/A | N/A | N/A | N/A | 15.21 ± 7.8 years | N/A | N/A | N/A | |
| 39 | 0 (0%) | 1479 women 100% |
46± 9.3 years |
CD4 count > 500 75 (34%) ≥200 and < 500 95 (44%) < 200 47 (22%) |
Plasma HIV RNA, Undetectable 92 (42%) < 10,000 73 (34%) ≥10,000 52 (24%) |
N/A | Yes 40 (15%) |
Diabetes Hypertension |
N/A | N/A | N/A | N/A | |
| 40 | 140 (81.0%) were known to be GBM | 0 (0%) | 47 (42–56) years | 632 (434–813) cells/ µL | Undetectable HIV viral load (<50 copies/mL), yes 153 (90.5%) | CD4 and CD8 T-cell | Yes 28 (19.4%) |
N/A | 6± 4 years | N/A | N/A | Among 98 individuals with 2 TE assessments,median time between SVR12 and post-DAA TE was 0.2 years (IQR: 0.1–0.6 years) | |
| 41 | 58 (68.2%) 37 (82.2%) |
27 (31.8%) 18 (17.8%) |
53 (47–56) years 51 (47–57) years |
412 cells/µL | N/A | N/A | 54 (63.5%) | Overweight = 20 (23.5%) | ˃9 years | N/A | N/A | N/A | |
| 42 | 159(79%) | 21% | 47 ± 9 years | 591 ± 254 cells/μL | N/A | Chronic elevation of ALT, AST and g-GT levels was present in 27% (n = 55), 19% (n = 38) and 49% (n = 98) of all patients, respectively. |
N/A | N/A | 13 ± 5 years | N/A | N/A | N/A | |
| 43 | 38 (100%) | 0 (0%) | 38 (35–42) years | 447 (375–604) cells/ μL | HIV RNA log10= 4.3 (3.6–4.9) HCV RNA log10 = 6.0 (5.3–6.8) |
ALT <2.5 3 ×ULN = 15 (40%) | 2 (5%) | Diabetes 2 (5%) |
6 years | N/A | N/A | N/A | |
| 44 | 75.2% | 24.8% | 37.0 years | 211cells/μL | N/A | N/A | N/A | N/A | ˃ 7 years | N/A | N/A | N/A | |
| 45 | 69.7% | 30.3% | 47.8 ± 6.4 | 564 (±309) cells/mm3 | N/A | N/A | Low-risk 916 (94.2%) High-risk 56 (5.8%) |
Obesity 44 (4.6%) Diabetes 11 (1.1%) |
˃8 years | N/A | N/A | being HCV cured were significantly associated with lower odds of having Advanced Liver Fibrosis. | |
| 46 | 65 men (69.8%) |
28 [30.2%] | 56.7 years | 564 cells/mm3 | HIV viral loads <50 copies/mL 76 (88.4%) Mean log HCV viral load =5.9 +/- 1.1(Mean +/- S.D) |
N/A | Yes 46 (49.5%) |
N/A | ˃ 14 years | N/A | N/A | N/A | |
3.2. Study Characteristics
The analyzed studies represent a global coverage with at least a representation from a major geographical region. The pooled data involved over 35,000 study subjects. The populations from all the included studies were mainly adults with a mean age of 40±6.4 years. Most of the studies were conducted with male gender domination though a fair number of studies were performed on females. We did not import low-quality articles. Fortunately, we did not have any. Analytically, 37 articles were of good quality (79.4%) and the other 10 articles were of fair quality (20.6%).
3.3. Analysis of Reported Etiologies
3.3.1. Etiologies Associated with Cirrhosis Progression
The analysis of various studies indicates that some factors are closely associated with fibrosis and cirrhosis progression in patients coinfected with HIV and HCV. Hepatitis C and HIV as long-term coinfection reported by various studies is a strong factor for the development of Cirrhosis [18-20, 23-28]. Additionally, high HIV viral load [29, 30], high HCV RNA load [17, 19], lower CD4+ count (<250/mm3) [20, 27, 29, 30], CD8 T-cell count, BMI [21, 22, 24, 25, 31, 32], older age [17, 19, 33, 34], high-risk alcohol consumption [17, 18, 21, 22, 24, 26, 27, 31, 35-37], chronic elevation of ALT, AST, APRI and Gamma-glutamyl Transferase (GGT) [17, 20, 23, 30, 34, 38-41], male gender [17-20, 29, 30, 33] and IV drug injection [17, 30, 36] have been reported as major etiological factors causing fibrosis and rapid progression of Cirrhosis in these patients. According to various studies conducted, these factors have been found to be statistically significant. Other factors singly reported by studies include caspase activity [19], higher sCD163 levels [39], low albumin concentration [33] and high Visceral Fat Area (VFA) [42], hypertension and diabetes [32], low serum levels of HDL in patients with ALT > 40 IU/L [41], cHCV mono-infection [26], unemployment, not currently receiving ART, currently receiving HCV treatment [31], exposure to certain anti- retrovirals including protease inhibitors and dideoxynucleosides and low serum cholesterol [23], mtDNA haplogroups cluster Others and V [43], and nonresponse to acute HCV therapy [21].
3.3.2. Other Suggested Etiologies
A significant difference was found in the level of bilirubin and hyaluronic acid in Cirrhosis progressing and non-progressing groups in Sandra et al.’s [20] study. Regarding molecular basis, polymorphism of PNPLA3 rs738409 genes [44], and IFNL SNP rs8099917 genes [45], higher levels of HLA-B18 [46] and IL-6 [47] have been reported to be associated with liver fibrosis in HIV/HCV coinfected patients. Moreover, advanced liver fibrosis was related to poor NK cell activity and elevated expression of the exhaustion/senescence marker PD1 in this population [48]. Foca et al. [30] reported that the occurrence of AIDs, FIB-4 class 3, kidney and cardiovascular diseases, and prolonged exposure to NRTIs were significant factors for outcome predictions. Conversely, a study [25] presents BMI, hyperlipidemia, alcohol abuse, HIV viral load, diabetes, and CD4 count as factors that are not associated with cirrhosis progression in HIV/HCV coinfected patients. Additionally, HCV genotype four has been identified as a protective factor for the development of liver cirrhosis [42]. The ideal cutoff value of VFA identified for the prediction of liver cirrhosis is 209 cm2 [42].
Moreover, several studies have identified factors associated with the development of hepatocellular carcinoma (HCC) [1, 5, 18, 26, 38]. While most groups did not show statistical differences, age, male sex, lower albumin levels, genotype 3, serum anti-HBc positivity, and a baseline liver stiffness greater than 40 were found to be positively associated with the incidence of Hepatocellular Carcinoma (HCC) [5, 18]. Additionally, achieving Sustained Virological Response (SVR) was linked to a lower incidence of HCC, although this association was not statistically significant [26]. Findings of a study focusing on AIGULr patients revealed that the baseline Liver Stiffness, Child-Turcotte-Pugh stage, and prior exposure to anti-HCV therapy before enrollment emerged as factors uniquely linked to liver-related mortality and/or transplantation [53].
SVR has been shown to play a protective role in liver-related morbidity and mortality [5, 9, 10, 22, 26, 38, 46]. Carrión et al. found that relative telomere length was lower in HIV/HCV co-infected individuals compared to those only affected by HCV after achieving SVR [28]. Furthermore, achieving SVR was associated with decreased odds of cirrhosis and fibrosis development [38, 46].
4. DISCUSSION
This review study was conducted on 47 articles to evaluate the risk factors of liver cirrhosis in HIV and hepatitis C coinfected patients. The results showed that HIV coinfection remarkably shortens the chance of survival of HCV patients in the cirrhosis stage. This effect is somehow dependent on age, gender, BMI, duration of HIV infection, and CD4 count.
One of the main causes of hospitalization and death in HIV-infected people is the complication of liver disease. Also one of the most frequent causes of advanced liver fibrosis to Cirrhosis in HIV patients is chronic hepatitis C [64]. HCV could be transmitted to about 30% of HIV-infected patients by shared transmission methods [65]. HIV coinfection was associated with fibrosis progress to Cirrhosis and some severe other liver complications [11, 66]. The study results indicated that coinfection of chronic hepatitis C and HIV was the main risk factor for cirrhosis. Results of a new meta-analysis study that has recently been published showed long-term HIV is one of the strongest pieces of evidence that contribute to HCC [67].
Based on the results of more than ten studies, it was indicated that elderly people and high-risk alcohol consumption were other essential reported risk factors for cirrhosis. Results of a similar study highlighted individuals' HIV and HCV co-infected who consumed higher alcohol are highly associated with liver complications [31, 68]. Older ages are highly associated with advanced fibrosis in HIV and HCV separately or coinfection [69-71]. Some longitudinal studies indicated higher levels of transaminases had a significant relationship with liver fibrosis progress among individuals with HIV-HCV coinfection [72, 73].
Results of four studies proved CD4+ less than 250 mm3 was significantly associated with liver cirrhosis, a study showed greater CD4 could decrease the probability of advanced fibrosis [31].
Few studies surveyed the impact of genetic factors as a risk factor among HIV and HCV co-infected. Results of a study indicated that rs738409 polymorphism is significantly associated with fibrosis progress and the results of another proved a significant relationship between PNPLA3 re738409 G allele and fibrosis progress [44, 74]. However, no significant association between the abovementioned genetic factors and liver complication advances was reported [75]. A significant association between the HLA-B18 allele and liver fibrosis advances was observed. It is stated that alleles could increase the risk of liver fibrosis complications [46].
Among HIV/HCV coinfected patients, HCV viral loads and undetectable plasma HIV-RNA (<50-200 copies/ml) were investigated as probable prognostic factors for liver fibrosis by a total of 31 papers. Across these studies, undetectable or suppressed HIV-RNA ranged from a low of 59% to a peak of 100% [44, 55]. Most studies found associations between higher HIV viral suppression rates among coinfected individuals and relatively better liver conditions. One study among 1479 women, of whom 235 (16%) were HIV/HCV coinfected, stated that in comparison of subjects with significant and non-significant liver fibrosis, the Plasma HIV RNA suppression rate was 42% vs 55%, respectively (p<0.001) [32]. Similarly, Oliver et al., in their study among 5985 HIV/HCV coinfected subjects, reported that the HIV viral load suppression was significantly lower in cirrhotic (19.8%) compared to non-cirrhotic patients (29%) [41]. This would indicate that higher HIV viral loads could be interpreted as possible poor prognostic factors for liver cirrhosis development. In addition, the HCV viral load varied among HIV/HCV coinfected patients between 4.9 to 6.2 (log10 copies/ml) [28, 29], and two papers found higher serum HCV RNA among patients with more fibrotic liver conditions, and one paper associated liver fibrosis progression with high serum HCV RNA [17, 20]. In contrast, Marco et al. reported that, among patients with higher liver stiffness, surprisingly, HCV RNA was relatively lower (5.79 vs 5.91 respectively), although their different finding was probably due to the usage of liver cirrhosis biomarkers instead of liver stiffness, that had led to high false positive cirrhosis rate among HIV/HCV coinfected patients [59].
The possible association between gender and liver fibrosis was investigated [17, 20, 59]. It was indicated that a higher level of liver stiffness would be associated with the male gender. Rodriguez et al. expressed that 77% of HIV/HCV coinfected patients who developed Cirrhosis were male [27]. One similar study found no gender-associated discrepancies among HIV/HCV-coinfected subjects in terms of the onset of Cirrhosis [76].
The possible effects of HIV/HCV coinfection on cirrhosis development onset were discussed in 13 papers. Among their findings, the onset of liver fibrosis ranged between 5 to 15.2 years. While one systematic review of 57 articles estimated that cirrhosis onset after 20 years of chronic HCV infection was 22% for the liver clinic series [77]. Therefore, it seems that HIV/HCV coinfection would shorten the onset of cirrhosis development. Rodríguez-Torres et al. in their study among 469 HCV mono or HIV coinfected cases, reported that HCV mono-infected compared to HIV/HCV coinfected cases had a significantly higher median risk of 42.0 vs. 32.0 years after infection for Cirrhosis, and specified that the median age of cirrhotic HCV mono-infected and HIV/HCV coinfection was 53.0 and 42.0, respectively [76].
High body mass index was another strong risk factor for liver cirrhosis; results of a study showed HIV-HCV coinfected patients had a higher chance (about six times) of advanced liver fibrosis [31]. Other studies worldwide, such as the American cohort of chronic HCV and a study in Egypt, reported obesity and overweighting as essential risk factors for liver fibrosis among chronic HCV patients [78, 79]. Also, visceral abdominal fat is considered an associated factor with the risk of liver stiffness. This factor was discussed in three included studies [33, 41, 42]. Oliver et al. stated that the percentage of subjects with a BMI of >30 was relatively higher among non-cirrhotic HIV/HCV coinfected cases (22.9%) compared to cirrhotic subjects (19.9%) [41]. One similar study also found an independent association between low albumin concentration and high visceral fat area with the probability of cirrhosis [42].
Finally, concerning the impact of achieving SVR on the development of HCC and the progression of liver cirrhosis, several reviewed articles indicated that eliminating HCV could positively impact the incidence of liver-related mortality and morbidity [9, 10, 18, 26] and reduce the likelihood of advanced liver cirrhosis and fibrosis [38, 46]. In a study involving 3,271 individuals diagnosed with HCC after achieving SVR, a decreasing trend in the incidence of HCC was observed among those with cirrhosis who experienced treatment failure, followed by cirrhotic patients who achieved SVR, non-cirrhotic individuals without SVR, and cirrhotic patients with SVR, respectively [80]. Eman et al. suggested that the heightened presence of hepatic progenitor cells expressing CK7 and CK19 could potentially contribute to the liver's ability to regenerate under these circumstances. Their research revealed noteworthy negative correlations between sustained virologic response (SVR) and both the stage of liver fibrosis and the expression of CK19 [81]. In their research, Poynard et al. observed that 49% of patients achieving SVR experienced a reduction in cirrhosis, yet 15 new cases of cirrhosis emerged among SVR patients with advanced fibrosis. Their longitudinal analysis over a 10-year period revealed that only 5% of cases showed regression of cirrhosis [82]. The other study also identified associations between younger age and higher platelet count with a greater probability of fibrosis regression, implying that early-stage cirrhosis might regress more readily than established cirrhosis, potentially influenced by portal hypertension.
CONCLUSION
Overall, the results showed that HIV coinfection greatly affected the severity of Cirrhosis in HCV patients. In summary, HIV coinfection alters the natural process of HCV-related Cirrhosis. This change depends on several factors, such as age and gender as demographic factors, so elderly people and men are more negatively affected by HIV and hepatitis coinfection. The duration of HIV infection and viral suppression status (or CD4 count) are also determining factors, so people who have had HIV for a long time and have a higher viral load are significantly at risk of advanced liver diseases. Also, people who consume alcohol and with high body indexes have a higher chance of advanced liver fibrosis. Collectively, HIV accelerates the progression of HCV-related liver disease, with its effect intertwined with the immune system. In order to investigate the effects of other risk factors such as IV drug injection, albumin concentration, high VFA, levels of HDL in patients with ALT, and exposure to certain antiretrovirals including protease inhibitors and dideoxynucleosides and low serum cholesterol more clinical studies are needed.
LIMITATION
There are some limitations of this study. First, only original and English articles were included. Also, online databases of PubMed, Web of Science, Scopus and Embase were searched for systematic review, it is possible that a relevant may exist that has not been indexed in these databases.
AUTHORS' CONTRIBUTIONS
(1) The conception and design of the study: Esmaeil Mehraeen, SeyedAhmad SeyedAlinaghi
(2) Acquisition of data: Arian Afzalian, Nazanin Janfaza, Ramin Shahidi
(3) Analysis and interpretation of data: Sanaz Varshochi, Reyhaneh Jashaninejad, Zahra Pashaei
(4) Drafting the article: Esmaeil Mehraeen, Marcarious M. Tantuoyir, Parinaz Paranjkhoo, Zohal Parmoon, Elnaz Shahmohamadi, Roghayeh Salmani, Parisa Matini, Pegah Mirzapour, Hooman Ebrahimi, Ali Moradi
(5) Revising it critically for important intellectual content: SeyedAhmad SeyedAlinaghi, Shayesteh Jahanfar
(6) Final approval of the version to be submitted: SeyedAhmad SeyedAlinaghi, Esmaeil Mehraeen, Shayesteh Jahanfar
LIST OF ABBREVIATIONS
| HIV | = Human Immunodeficiency Virus |
| HPCs | = Hepatic Progenitor Cells |
| SVR | = Sustained Virological Response |
CONSENT FOR PUBLICATION
Not applicable.
STANDARDS OF REPORTING
PRISMA guidelines and methodology were followed.
AVAIALABILITY OF DATA AND MATERIALS
All the data and supportive information are provided within the article.
FUNDING
None.
CONFLICT OF INTEREST
The authors declared no conflict of interest financial or otherwise.
ACKNOWLEDGEMENTS
The present study was conducted in collaboration with Khalkhal University of Medical Sciences, and Tehran University of Medical Sciences.
SUPPLEMENTARY MATERIAL
PRISMA checklist is available as supplementary material on the publisher’s website along with the published article.
Supplementary material is available on the publisher’s website along with the published article.


