All published articles of this journal are available on ScienceDirect.
Preclinical Assessment of a Cartridge-Based Flow-Through Assay for Determination of Adult CD4 T-Cell Count
Abstract
Background:
Despite the emphasis on viral load testing, current HIV testing guidelines consider CD4 T-cell count measurement as an important criterion for assessing disease progression, making decisions about anti-retroviral therapy regime changes, and treating HIV infected individuals with opportunistic infections. The CD4 counting by established methods (e.g., flow cytometry) presents challenges not only in resource-scarce settings due to cost and lack of skilled technicians but also in resource-rich areas where it is limited to centralized facilities.
Objective:
Current options for Point-Of-Care (POC) CD4 enumeration are few and labor-intensive, prompting the need for newer technological methods that can overcome the aforementioned challenges.
Methods:
The novel and patented flow-through cell counting assay (FTCA) described previously (Bystryak et al., 2019) was developed further into a point-of-care CD4 testing system using a disposable cartridge device and a portable imaging instrument. A pilot study with ~100 samples using this device was conducted to assess the validity of FTCA as a POC test for the measurement of CD4 count.
Results:
The FTCA signal was found to be linear over a wide range (17 - 1540 cells/μL) of CD4 T-cell concentration. The FTCA method also exhibits a strong agreement with flow cytometry, with very low bias (− 7 cells/μL) towards CD4 count measurement.
Conclusion:
The cartridge-based FTCA method has great potential to be a fully quantitative method with low complexity, portability, low-cost, and wide applicability in clinical practice.
1. INTRODUCTION
Human Immunodeficiency Virus (HIV) infection continues to be a major global health concern as the number of infected people increased from ~25 million in 2000 to ~38 million in 2018 [1]. However, the annual rate of global new HIV infections dropped from 2.8 million to 1.7 million during the same time period, and AIDS-related deaths have decreased from 1.4 million to 770,000, partly due to the increased number of infected people accessing antiretroviral therapy, from 576,000 in 2000 to 23.3 million in 2018. With no effective vaccine currently available, the development of a wide screening and identification assessment is needed in order to implement the appropriate intervention strategy. A decrease in CD4 T-lymphocyte count is associated with a compromised immune system [2-5], making it an important biomarker of disease progression in HIV infected individuals [6, 7]. The current guidelines of the World Health Organization (WHO) recommend initiation of Antiretroviral Therapy (ART) upon HIV diagnosis irrespective of CD4 T-lymphocyte level [8]. Early initiation of ART has shown to improve the quality of life and increase life expectancy in HIV infected individuals [9, 10]. Ata CD4 count of 500 cells/μL, ART initiation has shown to aid with viral suppression and reduce the risk of HIV transmission [11, 12]. The absolute number of CD4 T cells/µL of blood has critical prognostic and therapeutic implications and is used for both HIV staging and treatment decisions. Therefore, the WHO guidelines prioritize initiation of ART for individuals of all ages with severe or advanced HIV disease, characterized by a CD4 count of ≤350 cells/μL in adults, and a WHO clinical stage 3 or 4 HIV disease or a CD4 count of ≤750 cells/μL in children<5 years of age. During the first two years of treatment, both viral load testing and CD4 count measurements every 3-6 months are recommended [13]. In situations where viral load testing is unavailable, the updated guidelines strongly recommend CD4 counts as an important criterion for monitoring disease progression in HIV-infected patients and making critical decisions about treating HIV infected individuals with opportunistic infections and changes in ART regimes. In this regard, the CDC and NIH guidelines provide critical recommendations on prophylaxis of recurring and/or potentially life-threatening opportunistic infections in HIV positive patients with CD4 counts in the range of 100–200 cells/μL. Accordingly, the CD4 counts need to be monitored every 3–6monthsregardless of HIV plasma viral load levels in some cases [14].
CD4 counting by flow cytometry or FACS remains the gold standard in the HIV testing industry. Testing services in developed countries are offered at many centralized facilities using commercially available multi-platform flow cytometers like BD FACSCount, FACSCalibur (BD Biosciences), and EPICS XL (Beckman Coulter). These systems are expensive and require highly skilled technicians. A more cost-effective way in less developed areas is to use single platform systems dedicated for CD4 counting, such as FACS Count (BD Biosciences), CyFlow Counter (Sysmex Partec), Muse/Guava Auto CD4/CD4% system (EMD Millipore Sigma), and Apogee Auto40 (Apogee Flow Systems) [15, 16]. Apart from flow cytometry systems, there are two instruments for CD4 quantitation that are based on fluorescence cell imaging in a cartridge, namely Alere PIMA™ CD4 analyzer (Abbott Laboratories) [17, 18] and BD FACSPresto™ near-patient CD4 counter (BD Biosciences) [19]. The PIMA CD4 analyzer costs more than 5000 USD and provides agreeable CD4 counts with more sophisticated flow cytometers [20-28], with a low throughput of 3 samples per hour [29]. The FACSPresto measures absolute and percent CD4 T lymphocytes, and total hemoglobin (Hb) concentration. There are also some manual alternatives for CD4 counting that can be used in resource-limited settings. These methods are less expensive ($5-6 per test) but can be labor-intensive and challenging to implement [30]. One such method, the Dynal T4 Quant kit (Thermo Fisher Scientific) [16, 31] uses magnetic beads and requires an epifluorescent microscope, or even a light microscope, a hemacytometer, a vortex, a tube rocker, a timer, and a magnet, to perform cell isolation within 30 min [32]. Another system, the VISITECT® CD4 point-of-care test (developed by the BurnetInstitute, Melbourne, Australia) [33], is an instrument-free, semi-quantitative test based on lateral-flow technology and provides test results in 40 min.
Cartridge based systems using vertical flow technology have been developed for the point-of-care determination of WBC counts [34] and rapid testing of infectious diseases, STD's, and other conditions [35]. We have previously reported a novel method for point-of-care enumeration of CD4 T-cells using a patented Flow-Through Cell-Counting Assay (FTCA) [36, 37]. The FTCA is based on an immuno-concentration method and employs a special leukoreduction filter membrane to retain WBC's (including CD4 T-lymphocytes) [38]. The laboratory setup of the reported FTCA consisted of a controlled sample delivery system and optical imaging of the membrane for the quantitative colorimetric assessment of CD4 T-cell concentration. In this paper, we report recent developments of the FTCA system, including prototypes of the cartridge and optical imaging system. We also report a pilot study using these prototypes for preclinical validation of the FTCA for CD4 T-cell enumeration and correlation with the flow cytometry method.
2. MATERIALS AND METHODS
2.1. Materials
Custom purified mouse anti-human CD4 antibody, clone SK3, was bulk ordered from Becton Dickinson (Franklin Lakes, NJ). The purified antibody was custom conjugated with Alkaline Phosphatase (AP) by MOSS Inc. (Pasadena, MD) and supplied at a concentration of 1 mg/ml. Leukoreduction filter LRF10S was purchased from Haemonetics Corporation (Braintree, MA). DynabeadsCD3, DynabeadsCD14, and a magnetic separator rack were purchased from Invitrogen/Thermo Fisher Scientific (Waltham, MA).AP substrate (BCIP/NBT) solution and stabilizing diluent for ALP conjugates from MOSS Inc.; Levamisole solution from Vector Laboratories (Burlingame, CA); Muse® Human CD4 T Cell Kit from EMD Millipore Corporation (Billerica, MA); PE mouse anti-human CD14 from BD Biosciences (San Jose, CA) and absorbent pads were purchased from Bio-Rad Laboratories (Hercules, CA). All other reagents were purchased from Millipore Sigma (St. Louis, MO).
2.2. Blood Samples
Donor blood samples drawn in K3EDTA tubes were purchased from Biological Specialty Corporation (Colmar, PA) and used within 24 - 48 hours from the collection time. The blood samples were processed immediately upon receipt overnight. The total lymphocytes, CD4 T-lymphocytes, and monocytes were counted using a Guava EasyCyte flow cytometer (EMD Millipore). Samples with lower CD4 counts than normal were prepared either by (a) dilution of the whole blood with WBC-depleted autologous blood (Section 2.2.1.) or by (b) CD3 depletion of the whole blood (Section 2.2.2.). Overall~100 blood samples, including whole original blood and processed blood, were used in this study covering a wide range (17 - 1540 cells/μL) of CD4 T-cell concentrations.
Written consent for collecting blood samples was obtained by Biological Specialty Corporation from which such samples were acquired. Clinical samples were coded and unlinked to maintain patient privacy.
2.2.1. Dilution with WBC-depleted Blood
The whole blood was centrifuged in a clinical centrifuge for 10 min and half of the clear plasma from the top was collected in a separate tube. The remaining plasma, along with the buffy coat and half of the packed RBC under the buffy coat, was discarded. The clear plasma collected initially was mixed in a 1:1 ratio with the leftover packed RBCs to prepare WBC-depleted blood. Whole blood was mixed with autologous WBC-depleted blood in various ratios to obtain blood samples with various CD4 counts lower than that of the whole blood. This method also reduced the total WBC count in diluted samples without significantly affecting the hematocrit.
2.2.2. CD3 Depletion
CD3 T-lymphocytes were depleted using Dynabeads CD3, as described in the manufacturer’s instructions with some modifications. Briefly, 50 μL of Dynabeadswas added to 1 mL of undiluted whole blood and the sample was incubated with tilting and rotation for 30 min at 4 °C. After incubation, the sample was placed on a magnet for 1 min; the CD3-depleted supernatant blood was transferred to a new tube and the magnetic separation process was repeated for an additional 1 min to ensure complete separation of CD3-depleted blood from the Dynabeads. The supernatant was finally transferred to a second new tube and the remaining CD4 T-lymphocytes in this sample were counted by flow cytometry.
2.3. FTCA Protocol
As described earlier [36, 37], the FTCA method is based on measurement of the concentration of CD4 T-cells retained on a leukoreduction filter membrane after unbound RBCs have been washed away. In short, the FTCA method comprises of four steps: (1) mixing of the whole blood sample with CD4 antibody-AP conjugate, (2) controlled application of the reaction mixture on the membrane, (3) membrane washing to remove unbound reagents and cells, and (4) addition of AP enzyme-substrate and color development step on the membrane. A more detailed protocol is described below.
All reagents were equilibrated to room temperature and used according to manufacturers’ recommendations. The AP-conjugated anti-human CD4 antibody was diluted 250-fold (i.e.4μg/mL final concentration) with the stabilizing diluent for ALP conjugates. Nine μL of the diluted antibody-AP conjugate was mixed with 42 μL of blood sample and9 µL of Dynabeads CD14 magnetic beads in a 0.65 ml Eppendorf tube. The reaction mixture was incubated on a rotator for 12 min at room temperature and was then placed on a magnetic separator rack. After 3 min, with the tube on the magnetic rack, 50 μL of the supernatant reaction mixture was removed and applied onto the retainer membrane (pre-wet with 100 μL of 10 mM Tris, pH 7.4) through a 3.2 mm orifice of the insert in the FTCA cartridge (described in Section 2.4). Once the mixture was completely absorbed in the membrane (~15-20 s), the insert and the enclosure were removed and the membrane was washed twice with 1 mL of wash buffer (10 mM Tris, pH 9.5) added through the 10 mm orifice on the top of the FTCA cartridge. All reagents delivered on the membrane were allowed to soak completely before applying the next reagent. After washing, 100 μL of the AP substrate (BCIP/NBT solution) was applied over the sample spot on the membrane, and the FTCA cartridge was placed in the dark for color development. After 10 min, 200 µL of 0.1 N H2SO4 solution was added to stop the color development reaction, followed by 1 mL water to remove excess acid from the membrane. Test results were recorded and scored using a custom-built imaging system (described in Section 2.5).
2.4. FTCA Cartridge Prototype
Previously, we described a laboratory setup of the basic FTCA cassette comprising of a sample delivery tube held manually or by mechanical means over a WBC retainer filter membrane on an absorbent pad [36]. The use of a sample delivery tube/cylinder with a defined orifice size (4 mm diameter) and gently pressed over the membrane allowed a controlled sample application and an effective flow of the sample through a confined area of the membrane. These parameters were found to be crucial in our system for highly efficient retention of WBCs on the LRF10S membrane to obtain a more uniform and stronger signal compared to open/direct sample application on the membrane. However, the delivery of subsequent solutions (wash buffer, development solution, etc.) through the same tube resulted in higher background signal compared to when these solutions were delivered directly on the membrane, thus requiring the removal of the delivery tube after sample application. Based on these findings, we developed a disposable cartridge for performing FTCA. The current prototype of the FTCA cartridge shown in Fig. (1) is a multi-unit assembly comprising of four parts, (i) a removable insert or sample application port/device; (ii) a cartridge top; (iii) a cartridge bottom; and (iv) an enclosure, as shown in Fig. (1A). All parts were designed using SolidWorks software and 3D printed (stereolithography) using Accura Xtreme White high-resolution material build in 0.002” layers (Proto Labs, Inc., Maple Plain, MN). The characteristics of cartridge parts are described below:
The insert or sample application port/device (labeled 1) is a rounded funnel-shaped unit for sample delivery through a 3.2 mm diameter orifice (1 or 2 orifices) at the bottom of a flat stem. It is positioned on the cartridge top (labeled 2) in two shallow groves at opposite ends of the top. The insert is removed after the sample application step in the FTCA protocol. The cartridge top has a 10 mm diameter orifice with peripheral indentation underneath to hold a 12 mm diameter membrane disk (0.18 mm thick) in place. The membrane (labeled 3) is sandwiched between the cartridge top and an absorbent pad (labeled 4) contained in the cartridge bottom (labeled 5). In the current design (Fig. 1A), the upper and lower parts of the cartridge are held with mini-screws. The insert can be held manually over the cartridge top or secured firmly by enclosing the entire cartridge assembly in an enclosure (labeled 6) with an opening to access the insert for the sample application. The overall dimensions of a fully assembled cartridge with the enclosure are 45 mm x 40 mm x 40 mm. After sample application, the enclosure and insert are removed, and subsequent solutions are delivered through the 10 mm orifice in the cartridge top. The use of a removable sample application device (i.e., insert) over a fixed cartridge top allows sample delivery through a 3.2 mm orifice in the insert and limits sample application to a defined area (sample spot) on the membrane as mentioned earlier. After removing the insert, washing in “open” through the 10 mm opening of the cartridge top results in minimal background signal. It should be emphasized that the use of mini-screws and enclosure was necessary because all parts were 3D printed and were not suitable for assembly using a snapping mechanism. In the final design of the cartridge, however, a snapping mechanism will be used for holding all parts together, eliminating the need for mini-screws and enclosure (Fig. 1B). This assembly mechanism will be feasible because injection molding will be employed for the manufacturing of all cartridge parts. In our study, we used two types of inserts, (i) 1-hole insert, and (ii) 2-hole insert, which both works via the same principle and provided practically the same results as described in Section 3.1.
2.5. FTCA Imaging System
A prototype of a low-cost and portable imaging device was developed for signal measurement from the wet membrane within the cartridge immediately after the experiment and without requiring any manipulation or handling of the membrane. The general views of the imaging system are shown in Fig. (2A-C). At the end of the FTCA test procedure, the cartridge is simply placed into the optical imaging device from underneath (Fig. 2C) for a numerical readout (image score) of the signal via analysis of the spot intensity on the captured image (Figs. 2D and 2E). The can-shaped imaging system is enclosed in a 1/16” thick aluminum tube with dimensions of 3” (diameter) x 6” (height). The aluminum shell also serves as a heat sink and encloses a computer USB powered 2.0megapixel camera (labeled 2), LED ring (labeled 3), and internal light and dark signal intensity references on a plate (labeled 4). Inside the shell (Fig. 2A) is a manifold of upper and lower/base plate rings supported by long bolts. The power unit/voltage regulator (4 - 12 V) is enclosed within the shell and mounted on the upper plate (labeled 1). The camera with a 2.8 - 12 mm varifocal lens is mounted under the upper plate and a 12 LED ring is mounted at a lower level closer to the bottom plate (labeled 5). The internal light and dark references are mounted on a plate (labeled 4) between the LED ring and the bottom plate. The bottom plate has a slot underneath for the cartridge (Fig. 2C). The internal references and the test sample spot on the membrane lie at the same focal distance from the camera lens.
During signal measurement, the imaging system is placed over the cartridge (Fig. 2C) and an image is acquired in the Portable Network Graphics (PNG) file format (Figs. 2D and 2E). A custom-built scoring algorithm computes test scores from image analysis involving a two-stage calibration: (1) correction of measured sample intensity to compensate for changes in lighting and camera parameters (brightness, contrast, etc.), and (2) conversion of corrected sample intensity into an image score. The first correction is based on using the measured intensity of internal light and dark reference marks and is performed simultaneously with the measurement of the sample intensity. The second correction is performed only once for multiple experiments (i.e., for dozens or hundreds of samples) and can be repeated if necessary. In this case, the value of the dark internal reference is assigned to the darkest signal by measuring an external reference spot on the cartridge (from a sample with the highest CD4 concentration).


The intensity correction function that converts measured intensity to normalized (initial, standard) intensity is assumed to be linear. The conversion of measured sample intensity into an image score is computed assuming a linear scoring scale over the entire range of the normalized intensities of the light and dark references. The process of signal measurement from one cartridge to capture an image and generate readout as an image score takes less than a minute. The imaging system is a robust yet versatile system and can be used for extended periods of time without overheating or instability issues. The imaging system was used for obtaining image scores during FTCA assay optimization in the cartridge and for additional testing of approximately 100 blood samples in a pilot study, the results of which are described below.
3. RESULTS
3.1. Preparation of CD4 T-cell Specific Calibration Curve Using a Modified FTCA Protocol
In an earlier paper [36], we presented the CD4 calibration curve by the FTCA method using a biotin anti-CD4 antibody-streptavidin AP conjugate system. We reported that the LRF10S membrane was efficient in providing a signal specific for CD4 lymphocytes only and that there was a minimal contribution of monocytes. In the present study, using a crosslinked anti-CD4 antibody-AP conjugate, a noticeable difference in signal was observed for samples containing the same concentration of CD4 lymphocytes but different concentrations of monocytes. To check if this was due to the use of a different conjugate than used previously, the samples were also tested with biotin anti-CD4 antibody-streptavidin AP conjugate as described previously [36]. We also performed experiments using a modified FTCA protocol with samples after the monocyte depletion step (Section 2.3), and representative results with both antibody-AP conjugate systems are shown in Table 1.
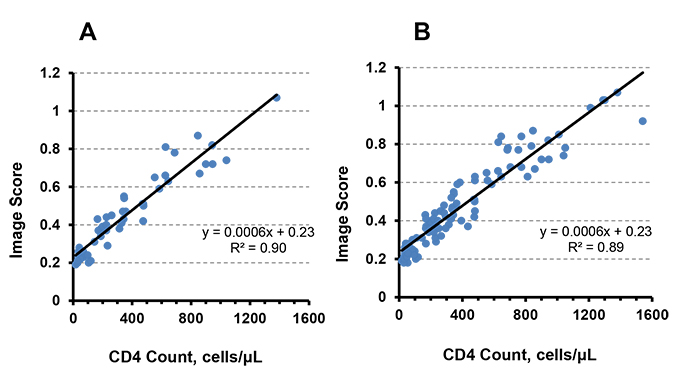
For both anti-CD4 AP conjugate systems, the image scores decreased with monocyte depleted samples, and therefore, the signal was specific to CD4 T-cell concentration. Using the modified FTCA protocol, the FTCA cartridge system was initially tested using a 1-hole insert, where one sample was applied at a time, and an image score was measured at the end. However, up to four cartridges were used in parallel for testing multiple samples simultaneously. Results from 47 samples using a 1-hole insert cartridge system are shown in Fig. (3A).
Similarly, 50 samples were tested using the 2-hole insert cartridge system. The calibration curve for the 2-hole system gave similar results (y = 0.0006x + 0.24, R2 = 0.89, data not shown) to the 1-hole system (y = 0.0006x + 0.23, R2 = 0.90). Since the 1-hole and 2-hole systems share the same operation principle and are identical in terms of their orifice diameter (3.2 mm), we combined the 1-hole insert and 2-hole insert cartridge data (a total of 97 samples) to get the calibration curve as shown in Fig. (3B). The combined calibration curve represents an overall distribution of samples covering a wide range (17 - 1540 cells/μL) of CD4 T-cell concentrations. All further results and discussion, including statistical analysis and correlation studies with a reference method, are based on the calibration curve from combined data.
| Conjugate System | Sample No. | Original Sample | Monocyte Depleted Sample | ||||
|---|---|---|---|---|---|---|---|
| CD4 Count* | Monocyte Count* | Image Score |
CD4 Count* |
Monocyte Count* |
Image Score |
||
| Biotin anti-CD4 antibody-Streptavidin AP | 1 | 520 | 669 | 0.63 | 515 | 6 | 0.57 |
| 2 | 307 | 669 | 0.55 | 305 | 4 | 0.41 | |
| 3 | 200 | 669 | 0.49 | 199 | 3 | 0.38 | |
| Anti-CD4 antibody-AP | 1 | 520 | 669 | 0.75 | 515 | 6 | 0.65 |
| 2 | 307 | 669 | 0.67 | 305 | 4 | 0.53 | |
| 3 | 185 | 284 | 0.51 | 153 | 0 | 0.38 | |
| 4 | 81 | 260 | 0.39 | 62 | 0 | 0.32 | |
3.2. Statistical Data Analysis
In order to estimate the clinical sensitivity and specificity of the FTCA method, a cut-off value, i.e., an FTCA signal at and below or above which samples will be considered positive or negative, respectively, needs to be established. The FTCA method sensitivity and specificity for the CD4 count threshold ([CD4]) at100, 200, 350 and 500 cells/μLwere evaluated using the ROC method described below.
3.2.1. ROC Analysis of Several Signal Cut-off Values
In this method, several signal cut-off(CO) values were selected for a specific threshold CD4 count ([CD4] = 100, 200, 350 or 500 cells/μL), and anoptimal CO value for clinical analysis was determined by receiver operating characteristic (ROC) analysis. The clinical specificity and sensitivity were determined based on the optimal CO value. For all samples, the FTCA signal (i.e., image score) was plotted vs. CD4 count measured by flow cytometry. Then, the FTCA calibration curve as shown in Fig. (3B) was prepared by using linear regression analysis,
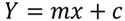 |
(1) |
where Y is the image score, and x is the sample CD4 count from flow cytometry. The CO value for the selected [CD4] was calculated from the calibration curve as CO [CD4]. Then, several CO values were selected within ± 20% of CO [CD4], and the most optimal CO value was determined by plotting the clinical sensitivity and specificity for various CO values by means of ROC analysis to achieve the best relationship between the clinical sensitivity and specificity.
The clinical sensitivity and specificity of the FTCA method were further calculated as,
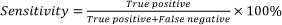 |
(2) |
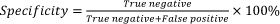 |
(3) |
where, True positive (TP), True negative (TN), False positive (FP) and False negative (FN) have usual meanings and are defined here as follows:
TP = sample CD4 count ≤[CD4]and image score≤CO [CD4].
TN = sample CD4 count>[CD4] and image score>CO [CD4].
FP = sample CD4 count>[CD4] and image score≤CO [CD4].
FN = sample CD4 count≤[CD4] and image score>CO [CD4].
A typical ROC analysis for [CD4] = 500 cells/μL is shown in Table 2. Similarly, ROC analysis was performed for [CD4] = 100, 200 and 350 cells/μL and the results are summarized in Table 3.
As shown in Table 2 and 3, the FTCA system, with a small number of samples, provides acceptable levels of sensitivity for all CD4 count thresholds, acceptable levels of specificity at [CD4] of 100 and 500 cells/μL, and moderate levels of specificity at [CD4] of 200 and 350 cells/μL. With further development of the technology and the use of a much larger clinical sample size, the clinical specificity of the FTCA method may be improved.
3.2.2. Correlation Studies
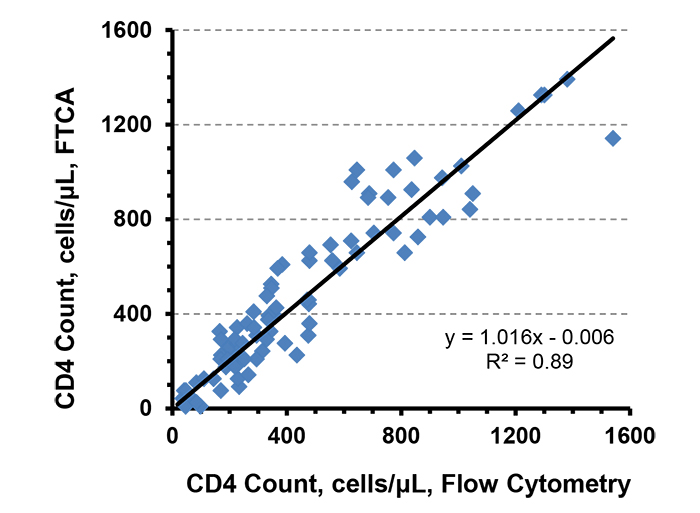
CD4 count determination by the FTCA method is based on measurement of the color intensity on the membrane rather than direct cell counting as in microscopy or flow cytometry. The color intensity is directly proportional to the concentration of CD4 T-cells in the sample. Flow cytometry, the current gold standard for CD4 count measurement, was initially used in order to obtain a preliminary understanding of the correlation of the FTCA method. The CD4 cell concentration calculated from the FTCA calibration curve (Fig. 3B) was plotted against the CD4 count measured by flow cytometry, as shown in Fig. (4). The correlation curve was linear over the entire range of CD4 concentrations studied, with a slope = 1.0, intercept = 0.006 and R2 = 0.9.
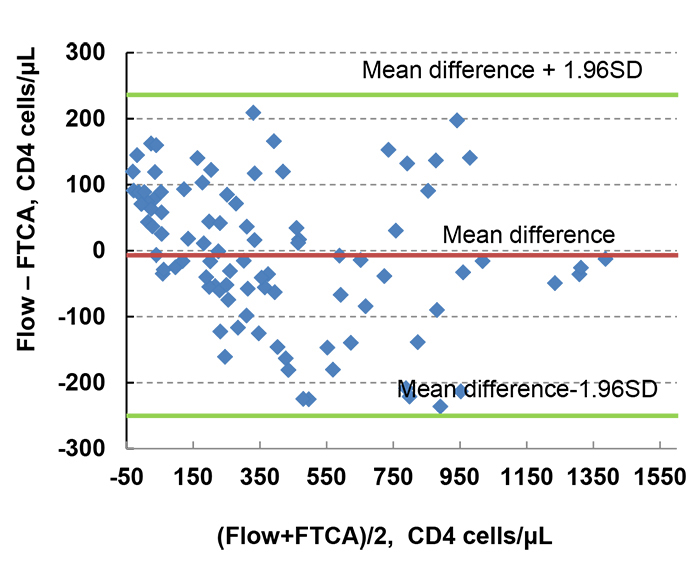
We also used Bland-Altman analysis to more accurately quantify the agreement between FTCA and flow cytometry, as shown in Fig. (5). The Bland-Altman comparison analysis predicts a bias of -7 cells/μL (95% confidence interval, -250 and 236 cells/μL), and all 97sample data are within 95% limits of agreement. These results indicate that FTCA has great potential to be a fully quantitative method.
| CO | TP | TN | FP | FN |
Sensitivity % |
Specificity % |
|---|---|---|---|---|---|---|
| 0.52 | 63 | 28 | 0 | 6 | 91 | 100 |
| 0.55 | 65 | 28 | 0 | 4 | 94 | 100 |
| 0.58 | 65 | 28 | 0 | 4 | 94 | 100 |
| 0.61 | 68 | 26 | 2 | 1 | 99 | 93 |
| 0.64 | 69 | 24 | 4 | 0 | 100 | 86 |
| [CD4] |
Optimal CO |
Sensitivity % |
Specificity % |
|---|---|---|---|
| 100 | 0.29 | 95 | 95 |
| 200 | 0.40 | 94 | 81 |
| 350 | 0.50 | 95 | 83 |
| 500 | 0.61 | 99 | 93 |
4. DISCUSSION
The basic principle of the FTCA method and selection of the retainer filter membrane for enumeration of CD4 T-cell count has been described previously in our earlier paper [36]. This paper describes the adaptation of the same basic concept towards the development of a point-of-care testing system, including a pilot study to demonstrate the feasibility of the FTCA cartridge and imaging prototype as a rapid test-system for CD4 counting. The key developments of the FTCA system are discussed here. In the current prototype of the FTCA cartridge, the use of a removable sample application device (i.e., insert) over a fixed cartridge top allows for a stronger and more uniform signal with minimal background [36]. We used two types of inserts, (i) 1-hole insert, and (ii) 2-hole insert; both types work via the same principle and give similar CD4 calibration curves. Using a 2-hole insert proves advantageous due to: (1) increased throughput, i.e., two samples can be run simultaneously; (2) a reference standard with known CD4 count can be run alongside the sample; (3) % CD4 for pediatric samples can be measured by running the same sample for CD4 and total lymphocyte counts using appropriate FTCA reaction protocols (to be published elsewhere).
The color intensity of the FTCA signal spot on the membrane is dependent on the CD4 count in the sample. The CD4 count in a sample may be estimated by visual comparison of the colored spot with a simple color paper chart, which may be quick and economical, but also s oversimplified and highly subjective. A more sophisticated, low-cost, portable, and readily available device like a glucose meter can be adapted for reflectance signal measurement, as described in our previous paper [36]. Some drawbacks of measuring reflectance signal include removal of the filter membrane from the cartridge (which may be contaminated) and careful drying of the membrane (at 37 °C for up to 15 min). These processes not only require careful manipulation and handling of the membrane, as reflectance is sensitive to membrane surface texture but also lengthen the testing time by about 20 minutes. To make signal measurement faster and more efficient without requiring any further handling of the membrane at the end of the assay, we developed an imaging system with image capture and analysis capabilities to accurately measure the FTCA signal from the wet membrane within the cartridge. Using a pilot study of approximately 100 samples, we showed that the image score of the FTCA signal is directly proportional to the CD4 count of the sample over a wide range of CD4 T-cell concentrations, 17 - 1540 cells/μL. The concentration of CD4 T-lymphocytes calculated from the analysis of the image score agrees well with the CD4 count measured by flow cytometry. Given its potential, we aspire to develop the imaging system as a standalone, portable, battery-operated device, with an integrated microprocessor chip, and a small LCD panel to display CD4 counts directly as part of a rapid diagnostic test. Future studies can be conducted with a larger number of blood samples to improve the power of the study.
In contrast to our previous study, we observed some monocyte interference of the FTCA signal using two different anti-CD4 AP conjugate systems, not specific to the new conjugate system used in this study. Although the reason for this discrepancy is unclear, the interference may be due to variations in the manufacturing process of the LRF10S membrane, causing an alteration of its properties [39].To overcome monocyte interference and to generate a signal specific for only CD4 T-lymphocytes with the current and future lots of the LRF10S membrane, we improved the FTCA protocol by including magnetic depletion of monocytes using anti-CD14 antibody magnetic beads, which does not alter the total reaction time of the FTCA protocol.
Results from the measurement of CD4 lymphocyte levels provide information that guides therapy and predicts disease outcome. In spite of viral load testing, current guidelines consider CD4 count as an important criterion for HIV disease management at various stages and ART decision making against opportunistic infections [8, 12, 14]. Although a few promising CD4 counting technologies have been developed [16, 40-44], they require complex systems (e.g., microfluidics), can be expensive, may struggle at advanced stages of development (e.g., Zyomyx CD4) [45], and sometimes provide erroneous results due to technical malfunctions (e.g., PIMA CD4 test) [26, 27, 46, 47]. The FTCA method is intended to be a semi-quantitative rapid point-of-care test that could be used in resource-limited settings, even by low skilled technicians, to monitor HIV disease progression at various stages of infection.
CONCLUSION
The FTCA method using a prototype of the cartridge and portable imaging system, offers a rapid test option for point-of-care determination of CD4 counts. The current system provides satisfactory sensitivity and specificity for measuring CD4 levels, allowing for appropriate treatment management (initiation, opportunistic infections, etc.) during HIV infection.
LIST OF ABBREVIATIONS
| AP | = Alkaline Phosphatase |
| ART | = Antiretroviral Therapy |
| BCIP | = 5-Bromo-4-Chloro-3-Indolyl-Phosphate |
| FACS | = Fluorescence-Activated Cell Sorting |
| FTCA | = Flow-Through Cell counting Assay |
| NBT | = p-Nitro Blue Tetrazolium chloride |
| POC | = Point-Of-Care |
| RT | = Room Temperature |
| STD's | = Sexually-Transmitted Diseases |
| WBC | = White Blood Cells |
AUTHORS’ CONTRIBUTIONS
SB, CA, KD, HZ and RPB conceived and designed the study. CA and RPB performed the assays. SB, HZ and RPB conducted the data analysis. RPB wrote the first draft.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by Essex Institutional Review Board, Inc(Lebanon, NJ), protocol No. B-012610-01.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Written informed consent was obtained from each contributor.
AVAILABILITY OF DATA AND MATERIALS
Raw data were generated at Allied Innovative Systems facility. Derived data supporting the findings of this study are available from the corresponding author [SB] on request.
FUNDING
This work was supported by the National Heart, Lung, and Blood Institute at the National Institutes of Health, grants R43 and R44HL097933.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We thank MykolaRabchevskiy of Gnosis Engineering LLC (Ewing, NJ) for contract fabrication of the imaging system, Michael Carden for routine laboratory technical assistance, and Lauren Sang for proofreading the manuscript.


