All published articles of this journal are available on ScienceDirect.
Effects of a 2-Year Supervised Exercise Program Upon the Body Composition and Muscular Performance of HIV-Infected Patients
Abstract
Background :
There is a lack of research investigating long-term effects of exercise training upon the body composition and muscle function in HIV-infected patients (PHIV). The study investigated the influence of a 2-year supervised exercise program on body composition and strength of PHIV under highly active antiretroviral therapy (HAART).
Methods :
A training program including aerobic, strength and flexibility exercises was performed by 27 PHIV (17 men/ 10 women; age: 48.7±7.0 years; HAART: 150.7±65.3 months) during 1 year and 18 PHIV (10 men/ 8 women; age: 50.6±5.2 years; HAART: 176.6±53.1 months) during 2 years. Body composition and knee isokinetic strength were assessed at baseline and at the end of each year of intervention.
Results :
Body composition remained stable along the whole experiment vs baseline (1-year - total muscle mass: Δ men=1.1%, P=0.21; Δ women=1.4%, P=0.06; trunk fat: Δ men=-0.1%, P=0.65; Δ women=-1.5%, P=0.45; 2 years - total muscle mass: Δ men=2.7%, P=0.54; Δ women=-1.9%, P=0.71; trunk fat: Δ men=4.4%, P=0.96; Δ women=10.0%, P=0.30). After 1-year, peak torque increased in men (Δ extension=4.2%, P=0.01; Δ flexion=12.2%, P=0.04) and total work reduced in women (Δ extension=-15.4%, P=0.01, Δ flexion=-17.5%, P=0.05). All strength markers remained stable vs baseline after 2 years of intervention (P>0.05). Only men showed significant reduction in the risk of disability due to sarcopenia (P=0.05) after 1 year of intervention, which remained stable after 2 years.
Conclusion :
Long-term exercise training preserved strength and muscle mass in PHIV under HAART. Exercise programs should be part of HIV therapy to prevent sarcopenia of this population along the years.
Trial Registration :
ACTRN12610000683033; UTN U1111-1116-4416.
INTRODUCTION
HIV-infected patients (PHIV) under highly active antiretroviral therapy (HAART) might exhibit collateral effects as increased proinflammatory profile [1, 2], hormonal impairment [2] and metabolic dysfunction [1-3]. Additionally to poor immunity [4] PHIV frequently exhibit greater accumulation of central adiposity and lipodystrophy [1, 5], chronic low-grade inflammation, and compromised growth hormone pulsatility (GH) [2, 6], among other consequences of the combination between HIV and HAART. Altogether these effects may contribute to the manifestation of an early “somatopause” in middle-aged PHIV [7], therefore increasing the risk of sarcopenia and premature physical disability [8].
On the other hand, regular physical exercise is considered as an effective strategy to counteract and prevent sarcopenia in frail populations, regardless of age, gender or clinical condition [9]. Physical exercise has been proven to improve muscle function and increase muscle mass [10-15], by contributing with the reduction of mitochondrial abnormalities [16] and proinflammatory cytokines (IL-6, TNF-α) [17], as well as increasing GH and IGF-1 levels [14, 18] or satellite cells differentiation [19]. Therefore, both aerobic and progressive resistance exercise are currently supported as beneficial for PHIV patients [10, 12, 18], and muscle strengthening is particularly considered by the patients as an important type of physical activity [20].
Accordingly, increases in strength and muscle mass due to exercise have been reported in PHIV [11, 13, 18, 21-23]. However, such beneficial responses were mostly observed after short periods of training (e.g. three to six months) [10-12, 18, 24]. In general, strength gains due to short term interventions are attributed not solely to an increase in muscle cross-sectional area, but also - and perhaps mainly - to neural adaptations [25]. Whether longer exercise programs would be capable to improve the muscle function in PHIV, and the extent to which this response would be related to sustained increase in muscle mass is yet to be defined.
Therefore, the purpose of this pre-post trial was to investigate the effects of a 2-year supervised exercise program upon the muscle strength and body composition of PHIV under HAART. We also assessed the prevalence of sarcopenia in these patients, based on cut-off points previously proposed [26, 27]. It has been hypothesized that the exercise program would induce gains in strength performance and muscle mass of PHIV, therefore reducing the prevalence and risk for disability due to sarcopenia.
METHODS
Subjects
Sixty-seven previously sedentary PHIV (48 men and 19 women) attending the infectious diseases outpatient clinic of the Pedro Ernesto Hospital of the University of Rio de Janeiro State, Brazil (HUPE-UERJ) initially volunteered for the study (57.5±6.0 years; 64.0±12.1 kg; 1.66±0.1 m). Eligible subjects should be older than 18 years-old and be under HAART treatment for at least 12 months. Exclusion criteria were: a) AIDS diagnosis; b) cerebral toxoplasmosis or any infectious diseases compromising the central nervous system; d) use of anabolic hormone therapy or ergogenic resources to gain muscle mass; e) cardiovascular, respiratory, bone, muscle, or joint problems that could limit physical function; f) less than 75% of attendance to the exercise sessions.
The PHIV were assigned into gender groups: HIV- men (HIV-M) and HIV-women (HIV-W). We considered as control reference the baseline data assessed before the exercise intervention. Due to ethical reasons, patients with less than 75% of attendance to the planned exercise sessions continued to exercise and therefore could not be regarded as drop-outs. In other words, only their data were not considered in the study due to low attendance.
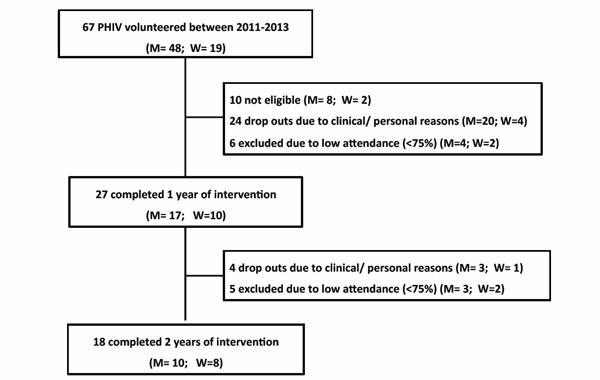
Flowchart of sample selection.
Baseline and relative changes (∆%) in clinical conditions and body composition in men.
| Variables | 1 Year (n=15) | 2 Years (n=8) | ||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | ∆% Baseline vs 1 Year |
P-Value | Baseline | ∆% Baseline vs 1 Year |
∆% 1 vs 2 Years |
∆% Baseline vs 2 Years |
P-Value | |
| Age (years old) | 47.0 (7.5) | − | − | 48.8 (5.7) | − | − | − | − |
| Time of HIV infection (months) | 187.2 (75.1) | − | − | 217.5 (72.1) | − | − | − | − |
| Time of HAART (months) | 169.7 (71.2) | − | − | 198.0 (59.8) | − | − | − | − |
| T CD4+ (cell/mm³) | 663.8 (311.2) | 17.7% | 0.51 | 767.9 (336.5) | 18.8% | -12.7% | 12.7% | 0.71 |
| Body Composition | ||||||||
| Body weight (kg) | 69.2 (10.4) | 3.0% | 0.17 | 66.1 (6.9) | 7.5% | 2.1% | 9.7% | 0.57 |
| BMI (kg/m2) | 22.8 (2.8) | 2.6% | 0.24 | 22.6 (2.0) | 2.2% | 2.5% | 4.0% | 0.92 |
| SMM (kg) | 51.8 (3.8) | 1.1% | 0.21 | 51.7 (2.0) | 2.0% | 0.7% | 2.7% | 0.54 |
| ASM (kg) | 24.2 (2.9) | 1.5% | 0.20 | 24.4 (1.3) | 1.9% | 2.0% | 3.9% | 0.75 |
| Body fat (%) | 21.9 (7.8) | 12.1% | 0.48 | 21.7 (8.2) | 1.8% | 5.8% | 7.1% | 0.73 |
| Trunk fat (%) | 27.7 (11.1) | -0.1% | 0.65 | 28.8 (12.1) | 2.4% | 3.8% | 4.4% | 0.96 |
| Fat mass (kg) | 15.4 (6.9) | 4.5% | 0.55 | 16.9 (4.9) | -6.3% | 7.9% | 0.6% | 0.78 |
| BMD (%) | 1.06 (0.1) | 0.1% | 0.90 | 1.03 (0.0) | 0.5% | 1.1% | 1.6% | 0.85 |
| Muscle Mass Indexes | ||||||||
| ASM index (kg/m2) | 7.8 (0.7) | 3.1% | 0.21 | 7.9 (0.2) | 3.3% | 1.3% | 4.5% | 0.35 |
| SMM index (kg/m2) | 8.4 (0.8) | 5.6% | 0.07 | 8.5 (0.3) | 3.4% | -2.4% | 0.8% | 0.54 |
Baseline values shown as mean (standard deviation) and variations as average percent delta (∆%). BMI: body mass index, ASM: appendicular skeletal muscle mass, SMM: total skeletal muscle mass, BMD: bone mineral density, ASM index: Appendicular skeletal muscle mass index adopted for sarcopenia cut-off points (Baumgartner et al. [26]), SMM index: Total skeletal muscle mass index adopted for sarcopenia cut-off points (Janssen et al. [27]).
Baseline and relative changes (∆%) in clinical conditions and body composition in women.
| Variables | 1 Year (n=10) | 2 Years (n=7) | ||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | ∆% Baseline vs 1 Year |
P-Value | Baseline | ∆% Baseline vs 1 Year |
∆% 1 vs 2 Years |
∆% Baseline vs 2 Years |
P-Value | |
| Age (years old) | 50.3 (5.3) | − | − | 52.0 (4.4) | − | − | − | − |
| Time of HIV infection (months) | 157.3 (68.2) | − | − | 188.0 (55.6) | − | − | − | − |
| Time of HAART (months) | 121.3 (43.4) | − | − | 148.0 (24.7) | − | − | − | − |
| T CD4+ (cell/mm³) | 724.8 (393.2) | 1.9% | 0.82 | 786.2 (307.2) | 10.8% | -6.9% | 4.2% | 0.98 |
| Body Composition | ||||||||
| Body weight (kg) | 56.2 (10.4) | 1.3% | 0.09 | 59.4 (10.7) | -0.2% | 1.6% | 1.4% | 0.48 |
| BMI (kg/m2) | 23.1 (3.8) | -1.5% | 0.54 | 24.1 (3.7) | -2.9 | 1.6% | -1.2% | 0.60 |
| SMM (kg) | 36.9 (3.8) | 1.4% | 0.06 | 38.7 (4.3) | -1.4% | -0.4% | -1.9% | 0.71 |
| ASM (kg) | 16.3 (2.0) | 0.4% | 0.71 | 17.0 (2.2) | -1.4% | 0.5% | -0.9% | 0.68 |
| Body fat (%) | 30.5 (7.5) | -1.0% | 0.59 | 30.9 (7.5) | 3.0% | 6.7% | 9.6% | 0.29 |
| Trunk fat (%) | 33.3 (8.7) | -1.5% | 0.45 | 34.3 (7.5) | 2.9% | 7.4% | 10.0% | 0.30 |
| Fat mass (kg) | 16.8 (8.3) | 0.6% | 0.80 | 18.2 (8.3) | 3.0% | 9.1% | 11.9% | 0.24 |
| BMD (%) | 1.01 (0.1) | -0.0% | 0.69 | 1.01 (0.1) | -2.1% | 0.0% | -2.0% | 0.17 |
| Muscle Mass Indexes | ||||||||
| ASM index (kg/m2) | 6.5 (0.7) | 0.4% | 0.78 | 6.6 (0.8) | -0.8% | 0.5% | -0.3% | 0.98 |
| SMM index (kg/m2) | 6.9 (0.8) | 0.5% | 0.71 | 7.1 (0.9) | -0.8% | 0.3% | -0.4% | 0.99 |
Baseline values shown as mean (standard deviation) and variations as average percent delta (∆%). BMI: body mass index, ASM: appendicular skeletal muscle mass, SMM: total skeletal muscle mass, BMD: bone mineral density, ASM index: Appendicular skeletal muscle mass index adopted for sarcopenia cut-off points (Baumgartner et al. [26]), SMM index: Total skeletal muscle mass index adopted for sarcopenia cut-off points (Janssen et al. [27]).
Baseline values and relative change (∆ %) of muscle performance markers (1 year, n=25; 2 years, n=13).
| 1 Year | HIV-M (n=15) | HIV-W (n=10) | ||
|---|---|---|---|---|
| EXT | FLX | EXT | FLX | |
| Peak Torque (N.m-1) | 153.5 (36.4) | 88.1 (28.8) | 119.1 (20.3) | 58.5 (8.7) |
| Baseline vs 1 year (∆%) | 4.2 * | 12.2 * | -2.6 | -6.3 |
| P-value | 0.04 | 0.04 | 0.52 | 0.31 |
| Average Power (watts) | 88.2 (23.9) | 45.7 (16.5) | 70.9 (14.0) | 34.8 (6.5) |
| Baseline vs 1 year (∆%) | 0.5 | 8.2 | -9.0 | -12.7 |
| P-value | 0.34 | 0.14 | 0.10 | 0.09 |
| Total Work (J) | 1475 (380.6) | 790.4 (271.9) | 1183 (147.7) | 613.1 (104.8) |
| Baseline vs 1 year (∆%) | 3.8 | 17.4 | -15.4 * | -17.5 * |
| P-value | 0.33 | 0.11 | 0.01 | 0.05 |
| 2 Years | HIV-M (n=7) | HIV-W (n=6) | ||
| EXT | FLX | EXT | FLX | |
| Peak Torque (N.m-1) | 179.6 (29.8) | 93.6 (22.3) | 117.9 (18.0) | 54.9 (8.1) |
| Baseline vs 1 year (∆%) | -0.8 | 9.9 | -1.6 | -1.4 |
| 1 year vs 2 years (∆%) | 3.2 | -6.8 | 0.7 | 6.4 |
| Baseline vs 2 years (∆%) | 1.1 | -2.2 | -1.3 | 5.1 |
| P-value | 0.95 | 0.74 | 0.97 | 0.78 |
| Average Power (watts) | 109.4 (20.9) | 59.3 (17.8) | 70.2 (12.1) | 30.1 (7.0) |
| Baseline vs 1 year (∆%) | -7.3 | -1.6 | -9.7 | 1.0 |
| 1 year vs 2 years (∆%) | 0.9 | -3.5 | 2.0 | 4.9 |
| Baseline vs 2 years (∆%) | -7.0 | -7.3 | -7.6 | 5.6 |
| P-value | 0.69 | 0.54 | 0.47 | 0.90 |
| Total Work (J) | 1826 (331.2) | 992.2 (342.4) | 1194 (166.3) | 539.6 (119.9) |
| Baseline vs 1 year (∆%) | -7.2 | 2.5 | -15.1 | -4.8 |
| 1 year vs 2 years (∆%) | 1.6 | 16.4 | 4.0 | 5.2 |
| Baseline vs 2 years (∆%) | -6.5 | 26.5 | -12.0 | 0.2 |
| P-value | 0.76 | 0.84 | 0.22 | 0.84 |
Baseline data are shown as absolute values (±standard deviation) in bold and relative changes are summarized as average percent delta. *: Significant difference compared to baseline (P ≤ 0.05). HIV-M: men and HIV-W: women. EXT: Knee Extension; FLX: knee Flexion.
Patients classified with moderate or high risk to physical disability due to sarcopenia according to the SMM index (Janssen et al. [27]) and as sarcopenic or non-sarcopenic according to the ASM index (Baumgartner et al. [26]) after 1 year and 2 years of supervised exercise intervention.
| 1 Year (n=25) | Group | Moderate Risk | High Risk | χ2 | P-value | Sarcopenic | Non Sarcopenic | χ2 | P-Value |
|---|---|---|---|---|---|---|---|---|---|
| Baseline | HIV-M | 8 | 7 | - | - | 3 | 12 | - | - |
| HIV-W | 3 | 1 | - | - | 1 | 9 | - | - | |
| 1 year | HIV-M | 10 | 2 | 2.7 | 0.05* | 3 | 12 | 0.0 | 1.00 |
| HIV-W | 3 | 1 | 0.0 | 1.00 | 0 | 10 | 1.0 | 0.30 | |
| 2 Years | Group | Moderate Risk | High Risk | χ2 | P-value | Sarcopenic | Non Sarcopenic | χ2 | P-Value |
| Baseline | HIV-M | 4 | 4 | - | - | 0 | 8 | - | - |
| HIV-W | 3 | 0 | - | - | 0 | 7 | - | - | |
| 1 year | HIV-M | 7 | 1 | 2.6 | 0.10 | 1 | 7 | 1.0 | 0.30 |
| HIV-W | 1 | 1 | 1.8 | 0.17 | 0 | 7 | 0.0 | 1.00 | |
| 2 years | HIV-M | 4 | 4 | 0.0 | 1.00 | 0 | 8 | 0.0 | 1.00 |
| HIV-W | 1 | 1 | 1.8 | 0.17 | 1 | 6 | 1.0 | 0.29 |
χ2: Chi-square. *: Significant difference compared to baseline (P ≤ 0.05). HIV-M: men, HIV-W: women. Cut-off points for detecting sarcopenia using the ASM index - 7.26 kg/m2 for men and 5.45 kg/m2 for women Cut-off points related to moderate and high risk of disability from sarcopenia using the SMM index - for men: 8.51-10.75 kg/m2 and ≤8.50 kg/m2, respectively; for women: 5.76-6.75 kg/m2 and ≤5.75 kg/m2, respectively.
Fig. (1) illustrates sample selection and drop-outs throughout the study. From the 67 initial volunteers, 10 were considered as not eligible according to exclusion criteria, since they exhibited cardiovascular disease (1 man), intermittent claudication (1 man), neurologic problems due to recent toxoplasmosis (3 men), or poor compliance to HAART (3 men and 2 women). After two years, 28 PHIV (23 men and 7 women) dropped out due to specific clinical and personal issues as work activities, disease, surgeries, motivation (i.e. family, travelling, reduced mood), or difficult access to the exercise facilities (i.e. traffic or long distance). Data from another 11 subjects (7 men and 2 women) were excluded from the analysis due to insufficient attendance (< 75% of exercise sessions), but these patients continued to exercise. In brief, 27 patients were assessed after 1-year experiment (17 men and 10 women; 48.7±7.0 years) and from these, 18 patients completed two years of intervention (10 men and 8 women; 50.6±5.2 years).
The study protocol was approved by institutional ethics committee and all volunteers gave informed written consent before participation in the study, according to the recommendations of the Helsinki Convention.
Experimental Design
The supervised exercise program took place between 2011 and 2013 and included aerobic, strength and flexibility exercises at the University of Rio de Janeiro State (UERJ) facilities. Participants were individually supervised by their physician assistants, in order to control their immunological status and drug therapy. The supervised exercise program took place three times per week, either in the morning or afternoon, according to patients’ best convenience. Training sessions occurred in all months of the year, except for a vacation interval from mid December to mid January. During this period the patients were recommended to keep physically active, by performing light tasks as walking or cycling. As part of the exercise program evaluations, all PHIV had their body composition and strength routinely assessed every year. Data were obtained in two visits to the laboratory, always in the morning period (8:00 to 11:00 a.m.). In the first visit the body composition was assessed by means of dual energy X-ray absorptiometry (DXA). After 24 to 72 h patients performed the isokinetic strength tests.
Body Composition Assessment
The DXA was used to assess whole and regional body composition (Hologic QDR 4500, HologicTM, Bedford, MA, USA). The DXA allows assessing body composition changes due to long-term infection and HAART, as the uneven distribution of fat, osteopenia and reduced skeletal muscle mass. The equipment was calibrated according to manufacturer instructions. Subjects were scanned in the supine position using standard protocols. Scans were performed in high resolution and analyzed by the same trained technician. Principles underlying DXA analyses were described elsewhere [28].
Isokinetic Strength Assessment
The strength of lower limbs was measured using a Biodex System 4 PRO isokinetic dynamometer (BiodexTM Medical Systems Inc., Shirley, NY). Calibration of the equipment was performed according to the manufacturer's specifications before every testing session. Prior to the tests the participants performed a specific warm-up in the isokinetic dynamometer (15 repetitions with angular velocity fixed at 120°.s-1).
The maximal isokinetic force protocol involved knee extensors and flexors in concentric-eccentric muscle action with the dominant limb. The range of motion varied between 0° to 90° with execution speed fixed in 60°.s-1. Subjects were verbally encouraged to perform maximal effort during 3 sets of 10 repetitions with 120 s intervals between sets. Peak torque, power output and total work were measured as indicators of force production and muscular resistance capacity. Percent delta (∆%) was used to calculate variations in each variable over the years. All tests were considered as valid only when the coefficient of variation between sets was lower than 15% as described elsewhere [29].
Sarcopenia Cut Off Points
The lean mass (Kg) of legs and arms was used to calculate the appendicular skeletal muscle mass (ASM). Cut-off values for sarcopenia proposed by Baumgartner et al. [26] in each sex were defined as values two standard deviations below sex-specific means from reference data for young adults aged 18-40 years obtained in the Rosetta Study [30]. Cut-off points considering the ratio between ASM and height squared were of 7.26 kg/m2 for men and 5.45 kg/m2 for women were adopted to classify individuals as sarcopenic. Janssen et al. [27] normalized the total skeletal muscle mass (SMM) for height (muscle mass (kg)/height (m)2) and termed it as SMM index. Cut-off points of 5.76-6.75 and ≤5.75 kg/m2 were reported to denote SMM index related to moderate to high risk of disability from sarcopenia in women. The corresponding values for men were 8.51-10.75 and ≤8.50 kg/m2.
These indices were created considering that the functional capacity related to a given amount muscle mass depends on individual body dimensions. Therefore, in Baumgartner and Janssen indices the muscle mass (appendicular and total, respectively) are related to height squared. The specific meaning of these indices also differ - while Baumgartner et al. [26] proposed cut off points to identify sarcopenia, the major concern of Janssen et al. [27] was to classify the risk for disability due to such important loss of muscle mass.
Exercise Intervention
Typically, participants trained three times per week in 60 to 90 min sessions including aerobic, resistance and flexibility exercises, as described elsewhere [11]. Aerobic training was performed on cyclo-ergometer or treadmill for 30-40 min at moderate intensity [50-te] to avoid possible exercise-induced immunodepression [31]. Resistance training included 8 to 10 single and multi-joint exercises (free weights and machines) for the upper and lower body, performed with 2 to 3 sets of 12 to 15 repetitions and load corresponding to 80-90% of 12 repetition maximum. The workload was adjusted every two months as described elsewhere [11, 32]. At the end of the sessions participants performed stretching exercises for the major joints using the static method [2 sets of 6 to 8 exercises with 30- to 8 exercises with 30-s duration]. All training sessions were supervised by exercise specialists on a one-to-one basis or in small groups (4-5 subjects per instructor).
Statistical Analysis
Normal distribution of data was confirmed by the Kolmogorov-Smirnov test and therefore results are presented as means ± standard deviations. Comparisons between groups were performed by repeated measures ANOVA followed by Bonferroni post-hoc verifications in the event of significant F ratios. Chi-square (χ2) was used to compare the pre vs post-intervention frequency of sarcopenia in the total sample and gender groups. All calculations were performed using the Statistica 7.0 software (StatsoftTM, Tulsa, OK, USA) and the probability level for statistical significance was set at P ≤ 0.05.
RESULTS
Program Adherence
An achieved statistical power of 0.793 for an effect size of 2.5 was obtained by performing a post hoc analysis (GPower version 3.0.10; Kiel, University of Kiel, Germany) based on the given sample size and predefined P value. No case of AIDS diagnostics or death was reported during the study, nor injuries or problems due to the exercise intervention. As aforementioned, 27 PHIV completed 1 year of intervention (17 men and 10 women) and 18 completed 2 years of this intervention (10 men and 8 women). The average adherence of those who remained in the study was 81% and 79% for 1 year and 2 years training groups, respectively. All PHIV were under HAART for at least 80 months (150.7±65.3 months).
Clinical Conditions and Body Composition
Baseline subjects’ characteristics and relative variation throughout 2 years of intervention with regard to clinical conditions and body composition are presented in Tables 1 and 2 for men and women, respectively. At baseline the viral load was undetectable (<50 copies/mL) for most patients. Only two men presented values above this cut-off point in 2011 and 2012 (2095 and 81 copies; 2518 and 3451 copies) and one man in 2013 (388 copies). All women showed undetectable viral load throughout the whole training period. T CD4+ count did not alter with the intervention 1 year later (men: ∆%=17.7, P=0.51, ∆%=18.8 and ∆%=-12.7% and women=0.82) and 2 years later (men: ∆%=12.7, P=0.71 and women: ∆%=4.2, P=0.98).
In regards to the muscle mass after 1 year of training program, slight albeit non significant increases have been observed in SMM (men: 1.1%, P=0.21 and women: 1.4%, P=0.06) and ASM (men: 1.5%, P=0.20 and women: 0.4%, P=0.71). The trunk fat remained quite stable in both men (-0.1%, P=0.65) and women (-1.5%, P=0.45). In the 2-years intervention group, SMM, ASM and body fat were maintained without expressive variations in both gender groups (Tables 1 and 2). Total muscle mass index (SMM index) applied to calculate sarcopenia cut-off points also remained stable over 1-year (men P=0.07; women: P=0.71) and 2-years of intervention (men P=0.14; women: P=0.86).
Strength Performance
Table 3 depicts markers of strength performance obtained in the isokinetic strength assessment. After 1 year of intervention, men presented significant increase in the maximal strength (peak torque) in relation to baseline, in both EXT and FLX (∆%=4.2%, P=0.01; ∆%=12.2%, P=0.04). Although average power and total work have increased in this group, they did not present significant difference in relation to baseline (average power - EXT: P=0.34, FLX: P=0.14; total work - EXT: P=0.14, FLX: P=0.11). Strength markers in women also showed to be stable along 1 year of training (peak torque- EXT: P=0.52, FLX: P=0.31; average power - EXT: P=0.10, FLX: P=0.09). Total work, however, significantly decreased in women that completed 1 year of intervention (EXT: ∆%= - 15.4%, P=0.01, FLX: ∆%= - 17.5%, P=0.05). Overall, after 2 years of intervention both men and women tended to maintain their muscle strength, despite of slight oscillations over the years (gains and losses) in most assessed variables.
Prevalence of Sarcopenia
Table 4 depicts the frequency of sarcopenia before and after exercise interventions. Albeit the non significant values obtained for the chi-square, there was a slight trend of reduction or stabilization of the number of individuals classified as sarcopenic as defined by the ASM index. Men showed a significant reduction in disability due to sarcopenia based on SMM index cut-off values after 1 year of exercise intervention (χ2=2.7, P=0.05).
DISCUSSION
The present study aimed to investigate whether body composition and muscle function could be improved in PHIV after long term exercise training. The main finding was that an overall exercise training routine performed over 2 years was efficient to maintain muscle mass and muscle performance in HIV-infected patients, therefore preventing sarcopenia and related risk of physical disability. Additionally, the immunological function reflected by CD4 count remained stable during the intervention.
A total of 29 randomized controlled trials could be found about exercise training for HIV-infected patients [21, 24, 33-57]. From these, eight trials applied resistance training [21, 33-39], eight applied aerobic exercise [50-57], and 11 applied a combination of aerobic and resistance exercises (concurrent training) [42-49, 58], while two compared the effects of resistance vs aerobic exercise [24, 40]. Sixteen studies measured both strength and body composition of participants [21, 24, 33-46], but only nine directly assessed the muscle mass [24, 33-39, 41]. As aforesaid, the majority of prior studies evaluating the effects of regular exercise in PHIV applied training protocols with relatively short duration. The duration of intervention programs specifically investigating the effects of exercise upon strength and body composition ranged from 6 to 16 weeks, but in approximately 20% of them it was shorter than nine weeks [21, 34, 38].
A number of short-term studies succeeded to demonstrate gains in both strength and/or muscle mass in response to exercise protocols with quite different characteristics [11, 12, 24, 33], including a previous controlled trial from our group that applied during 12 weeks exactly the same training protocol as used herein [11]. In the present study the muscle strength remained stable compared to baseline in the 2-year group, but increased significantly in the group that trained during 1 year. However, these gains were not concomitant to an increase in muscle mass, which might suggest that even in long term exercise intervention strength improvements would be mostly related to neural adaptations than to muscle hypertrophy [19]. This finding is consistent with premises from prior research suggesting that PHIV could improve muscle fitness in the same manner as sarcopenic elder individuals [17]. Additionally, it is feasible to suppose from our findings that even if the hypertrophy capacity is impaired in PHIV, this does not necessarily imply inability of the muscle to respond to exercise stimuli, at least in the sense of increasing the functional capacity and preserving the existing tissue.
Evidently, the existence of a non exercise control group would help confirming such hypothesis, particularly if non exercise controls had reduced their muscle mass and strength along the years in comparison with the trained groups. In any case, investigations in this regard are scarce. Actually, we could find only one study demonstrating a consistent decline of muscle mass in sedentary PHIV over five years compared to healthy controls [41], which reinforces our presupposition. Unfortunately, it was not possible to set a control group in the present trial and further investigation is necessary to provide a better insight on this matter.
Overall, data for isokinetic strength assessment indicated that muscle performance was maintained along the 2 years of supervised training in both sexes. However, women exhibited a slight decrease in total work (~15%) in the first evaluation (1 year), which was kept stable after 2 years of intervention. The peak torque can be defined as the highest muscular force output at any moment during a repetition, and represents muscle’s maximum strength capability. On the other hand, the total work reflects the muscle’s capability to maintain torque throughout the whole test. In a few words, the total work might be affected by both maximal strength and accumulated stress due to successive sets [29, 59]. Since the peak torque/ power output in men and women remained stable along the supervised training, it is feasible to speculate that the decrease in total work observed in females could be related to higher muscle fatigue during the test. Prior studies reinforce this premise, suggesting that there would be gender related effects of HAART upon hormonal levels, muscle fibers, and mitochondrial dysfunction [2, 60]. Evidently, further research is warranted to ratify this possibility, since additional reduction in total work in women was not detected after 2 years, despite the fact that HAART was not discontinued.
The preservation of strength and muscle mass in our sample had implications on pre vs post prevalence of sarcopenia and related risk for disability, as evaluated by indexes usually applied in elderly populations [26, 61]. A significant and fast decline in muscle mass has been reported in PHIV under HAART [41, 62] with consequent repercussion on the ability to perform tasks demanding important strength levels [7, 8]. An increased prevalence of sarcopenia along the years would be therefore expected among sedentary PHIV [8]. Our data suggested that regular exercise of moderate intensity and volume is, at least, capable to preserve the muscle mass in HAART treated PHIV over relatively long periods of time - the prevalence of sarcopenia in men and women remained stable during the 2-year follow up and decreased in men after 1-year intervention. To our knowledge, this is the first study to demonstrate that strength levels in PHIV may be increased or maintained due to exercise intervention longer than 1 year. Furthermore, it has been shown that this important functional outcome would be related to the preservation of muscle mass. Evidently, these findings should be taken into account in the context of long term treatment of PHIV - the beneficial effects of physical exercise seem to be consistent over the years, instead of transitory and limited to short periods.
The lack of a formal non exercise control is the major limitation of the present study. Actually, eligible patients that declined to participate of exercise training also refused to perform body composition and isokinetic assessments. For ethical reasons, those that fulfilled eligibility criteria could not be precluded to participate of the intervention. On the other hand, it is important to mention that previous research have adopted similar strategy of using preexercise data as a “control set” to investigate the effects of exercise upon muscle strength and mass in within-group trials [63]. Furthermore, follow ups investigating the effects of regular exercise for 1 year or more are very scarce - actually, most studies with concurrent training in HIV-patients are limited to 6-16 weeks, which limits inferences about the real impact of training on muscle mass and strength due to the major influence of gains related to neural adaptations [64-66]. On the other hand, the wasting syndrome is still a reality among HIV-infected patients, particularly when their come from low income social classes, which is the case of the majority of our patients. In this context, the present findings can be considered as clinically relevant despite the limitations of our protocol, since it has been originally demonstrated that long term exercise intervention is likely to preserve along the years the muscle mass, strength, and functionality of PHIV under HAART.
Additionally, it was not possible to control for changes in the antiretroviral type or dosage used by patients during the experiment. The HAART may include different drugs (i.e.: nucleoside transcriptase reverse inhibitors, protease inhibitors etc.) and depending on the type, dosage and time of utilization of a specific combination of them, different effects on mitochondrial function, muscle fiber and protein synthesis might be expected [3]. Also, analysis of other immunological markers (CD4:CD8 ratio, leukocytes count) would be useful to complement the immune markers presently assessed.
In conclusion, this pre-post intervention trial suggested that a supervised training program including aerobic, resistant, and flexibility exercises performed for 1 to 2 years would be capable to increase or maintain muscle performance and preserve muscle mass in HAART treated HIV-infected patients, with no apparently negative effects on the immunological function as reflected by the CD4 count. Long term physical exercise programs should be therefore considered as an essential part of HIV therapy, in order to prevent sarcopenia and preserve the functional autonomy of PHIV along the years. However, further research, particularly randomized controlled trials are warranted to ratify these preliminary results.
CONFLICT OF INTEREST
The authors confirm that this article content has no conflict of interest.
ACKNOWLEDGEMENTS
This work was partially supported by FAPERJ and CNPQ. We also thank the staff from the Interdisciplinary Nutritional Assessment Laboratory of the University of Rio de Janeiro State (LIAN) for conducting DXA exams.


