All published articles of this journal are available on ScienceDirect.
Mortality in Hospitalised HIV/AIDS Patients in a Tertiary Centre in Sub-Saharan Africa: Trends Between 2007 and 2015, Causes and Associated Factors
Abstract
Background:
With easy accessibility to combination Antiretroviral Therapy (cART), mortality amongst hospitalized HIV/AIDS patients needs to be described.
Objective:
We aimed at determining the trends, causes and factors associated with in-hospital mortality amongst HIV/AIDS patients in the Douala General Hospital.
Methods:
We retrospectively reviewed hospitalisation records of HIV/AIDS patients hospitalized in the medical wards of the DGH from 2007 to 2015. Four cause-of-death categories were defined: 1. Communicable conditions and AIDS-defining malignancies, 2. Chronic non-communicable conditions and non-AIDS defining malignancies’, 3. Other non-communicable conditions and 4. Unknown conditions. Logistic regression was used to determine factors associated mortality.
Results:
We analyzed 891 eligible files. The mean age was 43 (standard deviation (SD): 10) years and median length of hospital stay was 9 (interquatile range (IQR)4 - 15) days. The overall all-cause mortality was 23.5% (95% CI: 20.8% - 26.4%). The category - communicable conditions and AIDS defining malignancies represented 79.9%, of deaths and this remained constant for each year during the study period. Tuberculosis was the most common specific cause of death (23.9%). Patients who had two (OR=2.35, 95%CI: 1.35 - 4.06) and more than two (OR=4.23, 95%CI: 1.62 – 11.12) opportunistic infections, a haemoglobin level less than 10g/l (OR=2.38, 95%CI: 1.58 - 3.59) had increased odds of dying.
Conclusion:
In-hospital mortality is high amongst HIV/AIDS patients at the Douala general hospital. The category - communicable conditions and AIDS defining malignancies - is still the main underlying cause of death. We hope that our findings will help to develop interventions aimed at reducing in-hospital mortality.
1. BACKGROUND
Infection with the Human Immune Deficiency Virus (HIV) remains one of the world’s most significant public health challenges affecting an estimated 36.7million people of whom 70% (25.6 million) are in the WHO Africa region [1]. HIV is a leading cause of death worldwide and has claimed more than 35 million lives since its discovery over thirty years ago [2]. In 2016, one million people died from HIV-related illnesses of which 720,000 were from Africa [1]. In Cameroon, about 4.3% of the adult population are HIV positive [2]. At current rates there is a projected increase from 560,000 to 726,000 people living with HIV/AIDS by 2020 [3].
The pathogenesis of HIV infection is largely attributable to the decrease in the number of CD4 cell counts [3]. As the CD4 count falls, the patient becomes more susceptible to opportunistic infections with increased morbidity and mortality. Combination antiretroviral therapy (cART) suppresses the HIV viral load and improves immunological and clinical well-being [4-8].With scale up in the provision of cART, HIV infection has evolved from a fatal to a chronic disease. Hospitalization and mortality rates have dropped [9, 10]. However, in-hospital mortality still remains high in sub-Saharan Africa (SSA) ranging from 24.2% to 44% [11]. The causes of death have drifted from HIV-related to non-HIV-related conditions in resource rich countries [12-14]. In SSA, although the proportion of non-AIDS related deaths is rising, mortality is still largely due to AIDS-related conditions especially tuberculosis and bacterial infections [15-23].
In Cameroon, HIV care and treatment were expected to improve as free HAART has been available since 2007. As scale up is slow, current care is still largely focused on management of opportunistic infections. Awareness of a changing pattern of the disease and the causes of mortality in this setting is of importance for patient care and resource allocation [17]. We therefore aimed at determining the trends, causes and factors associated with in-hospital mortality of HIV infected patients from 2007 - 2015, in order to get a better picture of the changing phase of the HIV/AIDS pandemic in this setting.
2. MATERIALS AND METHODS
2.1. Study Design and Setting
This was a retrospective hospital-based study of HIV/AIDS patients admitted to the internal medicine wards of the Douala General Hospital (DGH) from 1st January 2007 to 31st December 2015.
The DGH is a 220-bed tertiary health institution situated in Douala, the economic capital of Cameroon which has a catchment population of about three million inhabitants. It is home to one of the centres of excellence in HIV/AIDS management in the region providing active care to about 2000 HIV infected individuals - the DGH accredited treatment centre (ATC). The centre employs a multidisciplinary team approach to HIV/AIDS management consisting of specialists in infectious diseases, internal medicine, neurology, chest medicine, as well as psychologists, pharmacists, primary care physicians, nurses and social workers [24]. The internal medicine wards (comprising all subspecialties) has a total capacity of 60 beds and an average influx of 250 patients monthly.
Management of patients with HIV is in accordance with the National Aids Control Committee guidelines which were adapted from the WHO treatment guidelines [25].
The HIV status of patients admitted to the hospital may be known if they were followed up in the DGH-ATC. Otherwise an HIV test is requested following clinical considerations after counselling. HIV diagnosis in this institution is made according to Cameroon National AIDS Control Committee guidelines by antibody detection on two successive samples using a third generation ELISA test BIOREX (Biorex Diagnostics Limited, Antrim, United Kingdom). If both are positive, a third sample is collected and tested using Genie III HIV-1/HIV-2 Assay (Bio-Rad Diagnostics, Marnes la Coquette, France) to specify either HIV 1 or HIV 2. A patient is declared positive for HIV if these three tests are positive. In case of any discordance, testing is done with Western blot (New LAV blot, Diagnostics, Pasteur, Marnes la Coquette, France).
In accordance with the National Aids Control Committee guidelines, patients with CD4+ T cell counts ˂ 500cells/µl were commenced on ART. This cut off was upgraded in accordance with the WHO guidelines of 2013. The initial cut offs were <200cells/mm3 from 2006-2009, <350cells/mm3 from 2010-2013, <500cells/mm3 from 2013-2015, and “treatment for all” in 2016. The hospital pharmacy dispenses ART and some prophylactic drugs as indicated, for opportunistic infections at no cost to the patients. Patient’s prescriptions are renewed monthly following a physicians’ visit.
2.2. Study Population and Data Collection
All medical files of HIV/AIDS patients hospitalized in the medical wards of the DGH during the study period were extracted. Excluded were files with insufficient data especially unrecorded death dates.
A structured, pretested data collection form was used to obtain information from patient files. HIV/AIDS patients were identified through the medical department’s in-patient registry. The medical records of these patients were then retrieved from the archives. Medical case files, nursing notes and discharge summaries were reviewed and relevant data obtained from eligible participants. Omitted data such as the most recent CD4 cell count and full blood count were traced from the participant’s out-patient medical record or from the laboratory registers when available (especially for those followed up in the DGH-ATC). Information retrieved were: Socio-demographic (gender, age, residence, marital status, employment status), clinical data (diagnosis at discharge or death, antiretroviral treatment regimens), laboratory (Haemoglobin level, CD4 cell count) and outcome of the patient (death, causes of death, discharge).
2.3. In-hospital Diagnosis
The criteria used to identify the main infectious disease/events were as described in the manual for prevention and management of HIV in Cameroon [25]. These were particularly for: tuberculosis, community acquired pneumonia, cerebral toxoplasmosis, candidiasis, pneumocystis pneumonia, cryptococcal meningitis, sepsis, chronic diarrhoea, Kaposi’s sarcoma, and lymphoma.
2.4. Assigning the Causes of Death
The cause of death was defined as the main condition that initiated the sequence of morbid events leading directly to death [26]. Two study team members (FM, MN) reviewed the participants’ medical records of death and independently determined the most likely cause of death. Concordance was then established. All cases were discussed and confirmed by internists and infectious disease specialists (HNL, HBM, MSD, YNM) before a final cause of death was assigned. This process was done even for those who had a cause of death noted in their files.
The causes of death in this study were then classified under four main categories namely; 1. Communicable diseases and AIDS-defining cancers: The primary cause of death was from one of the AIDs defining illnesses in HIV positive individuals at any immunological stage. The sub-category, ‘’other communicable conditions’’, was used for other non-AIDs defining infections in other organ systems not specifically mentioned in the previous group. When no specific cause of death could be assigned, but the cause of death was considered to be related to a communicable disease, for example because of the presence of fever, the term “communicable disease unspecified” was used. 2. Chronic non-communicable diseases/non-AIDS malignancies: Which included non-AIDS defining malignancies and chronic medical conditions (cardiovascular, digestive, renal disease etc,) 3. Other non- communicable diseases, where we included: acute renal failure, pulmonary embolism, etc.) 4. Unknown Category was assigned when no distinction could be made between a communicable or non-communicable cause of death.
The cause of death assigned to each adult patient was based on a listing of predefined causes of death, which was derived from the global burden of disease [17, 27, 28].
2.5. Ethical Considerations
Data collected was used solely for the purpose of this research. Confidentiality was respected as codes were used instead of participants’ names and only members of the research team had access to participants’ information. Authorization to carry out this study was obtained from the Faculty of Health Sciences of the University of Buea. Ethical approval was obtained from the Institutional Review Board of the Faculty Medicine and Pharmaceutical Sciences of the University of Douala (IEC-UD/474/02/2016/T). Administrative approval was obtained from the Director of the Douala General Hospital.
2.6. Statistics
The outcome of this study was mortality amongst hospitalised HIV/AIDS patients over time. Results were presented as count (percent), as mean (standard deviation) or as median (interquartile range) where appropriate. The chi2 test was used to test association between the outcome and explanatory variables as well as for trends. For analytical purposes, we further categorized variables with using clinical cut-offs and those with no a priori categorisation or clinical cut-off values were grouped in tertiles. We also regrouped calendar time in 3 years intervals to reduce the number of parameters to be analysed. To assess factors associated to hospital mortality, we used univariate logistic regression reporting Odd Ratios (OR) and their 95% Confident Intervals (CIs) and for this analysis, we used only variables that had at most 20% of missing values. We finally used all factors that showed at least a weak evidence of association (p value < 0.10) with the outcome alongside age, to build an exploratory multivariate logistic regression model (introducing all the variables in the same model). Results were analysed using STATA 13, and the threshold for significance was set at the level of 5%.
3. RESULTS
We identified 1081 HIV/AIDS patients from the in-patient register. Of these, 916 files were retrieved from the archives. A further 25 files were excluded because there was no recorded date of death or limited data. Included for analysis in this study were 891 files.
Characteristics of the study population: Baseline characteristics of the population are depicted in Table 1. Mean age was 43 (SD: 10) years. The proportions of men and women were 51.6% and 48.4%, respectively. Median (IQR) length of hospital stay was 9 (4 - 15) days. The highest number of admissions - 373 (41.8%) was observed between 2013 and 2015. On admission, 38.5% of the patients were on cART. Median (IQR) CD4 cell count was 92 (32 - 218) cells/mm3. Out of the 659 patients who had information on CD4 cell count, 72.2% had values lower than 200cells/mm3. Mean hemoglobin was 9.17 ± 3.61g/dl. Seventy five percent of the patients had at least one opportunistic infection at the time of admission.
| Characteristics | n (%) | Characteristics | n (%) |
|---|---|---|---|
| Mean age, N=891 | 43(±10) | Median hospital stay, N=877 | 9(4-15) |
| Gender, N=891 | Marital status, N=852 | - | |
| Male | 460(51.6) | Single | 283(33.2) |
| Female | 431(48.4) | Married | 505(59.3) |
| Calendar Year, N=891 | Divorced | 11(1.3) | |
| 2007-2009 | 210(23.6) | Widowed | 53(6.2) |
| 2010-2012 | 308(34.6) | Employment, N=877 | |
| 2013-2015 | 373(41.8) | Active service | 642(73.2) |
| Residence, N=855 | Unemployed | 194(22.1) | |
| Urban | 817(95.6) | Retired | 41(4.7) |
| Rural | 38(4.4) | ||
| HAART on admission, N=864 | Opportunistic infections, N=891 | - | |
| No | 531(61.5) | None | 215(24.1) |
| Yes | 333(38.5) | 1 infection | 474(53.2) |
| Haemoglobin, N=785 | 2 infections | 175(19.6) | |
| <10g/l | 494(62.9) | >2 infections | 27(3.1) |
| ≥10g/l | 291(37.1) | - | - |
| CD4, N=659 | - | - | |
| <200 cells/mm3 | 476(72.2) | - | - |
| 200-349 cells/mm3 | 102(15.5) | - | - |
| ≥300 cells/mm3 | 81(12.3) | - | - |
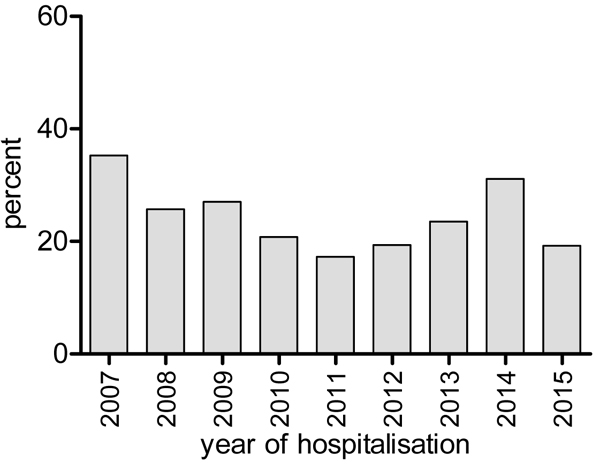
In-hospital mortality: Overall, all-cause mortality between 2007 and 2015 was 23.5% (95% CI: 20.8% - 26.4%). The highest proportion of mortality, 35.3% (18/51) was in 2007 and this decreased to 17.3% (18/104) in 2011 before rising again to 31.1% (37/119) in 2014. In 2015, the year with the highest number of admissions for HIV/AIDS, mortality dropped to 19.2% (30/156) (Fig. 1). However, there was no statistical evidence supporting a significant change in trends (P=0.31). Out of the 209 patients who died in hospitalisation between the years 2007 and 2015, the main cause of death was attributed to the category - communicable conditions and AIDS defining malignancies (79.9% = 167/209), followed by chronic non- communicable conditions and non-AIDS defining malignancies (8.1%), unknown cause (7.7%), and finally by other non-communicable conditions (4.3%). Throughout the study period, communicable conditions/AIDS defining malignancies were year by year, the most common category causing death (Fig. 2).
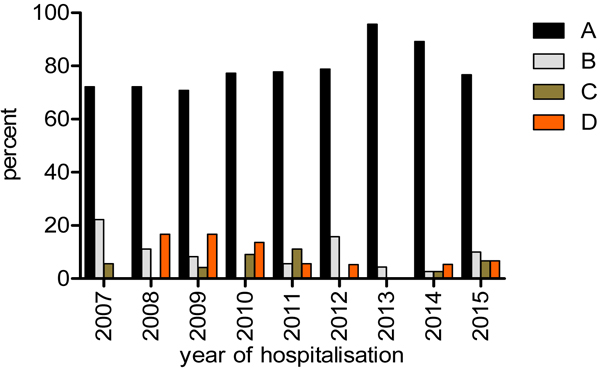
Legend: A=communicable conditions and AIDS defining malignancies, B=chronic non-communicable conditions and non-AIDS defining malignancies, C=other non-communicable conditions, D=unknown.
3.1. Cause Specific In-hospital Mortality
Tuberculosis was the most prevalent cause of death overall (23.9% = 50/209). This was closely followed up by Central Nervous System disease at 23.4% (49/209) - cerebral toxoplasmosis (21 cases), cryptococcal meningitis [16], and other meningitis [12]. The most common AIDS defining cancer was Kaposi’s sarcoma at 5.3% (11 cases) (Table 2).
| - | Total n (%) | Disease category | Total n (%) |
|---|---|---|---|
| Communicable/AIDS defining malignancies | 167 (79.9) | Chronic NC/non-AIDS Malignancy |
17 (8.1) |
| Tuberculosis | 50(23.9) | Non-AIDS-defining malignancies | 6(2.9) |
| Community acquired Pneumonia | 6(2.8) | Cardiovascular diseases | 1(0.5) |
| Pneumocystis pneumonia | 14(6.7) | Gastrointestinal/Hepatic diseases | 5(2.4) |
| Diarrhoeal disease* | 9(4.3) | Chronic Kidney disease | 5(2.4) |
| Cryptococcal meningitis | 16(7.7) | Other non-communicable | 9(4.3) |
| Cerebral Toxoplasmosis | 21(10) | Pulmonary embolism | 2(1) |
| Other Meningitis | 12(5.7) | Acute renal failure | 2(1) |
| Kaposi’s sarcoma | 11(5.3) | Other unspecified | 5(2.4) |
| Non-Hodgkin’s Lymphoma | 3(1.4) | Unknown | 16(7.7) |
| Other AIDS defining** Malignancies |
1(0.50) | - | - |
| Other communicable conditions | 16(7.7) | - | - |
| Communicable unspecified | 8(3.8) | TOTAL | 209(100) |
| *Chronic diarrhoea, amoebic dysentery**Invasive cervical cancer | Cardiovascular disease: Congestive heart failure | ||
In the chronic non-communicable and non-aids malignancy category were: Gastrointestinal/hepatic disease and chronic kidney disease at five cases each and non-aids malignancies, six cases.
Factors associated with mortality: In univariate analysis, calendar time, lengths of hospital stay, the number of opportunistic infections and a haemoglobin <10g/l were associated with mortality. When all these factors were included alongside age tertiles in multivariate analysis, patients aged 48 and above had higher odds of dying compared to those who were younger (OR=1.58, 95%CI: 1.00 - 2.49). Patients hospitalised in 2010-2012 and in 2013-2015 had reduced odds of dying compared to those hospitalised in 2007-2009 (OR=0.53, 95%CI: 0.33 - 0.87 and OR=0.57, 95%CI: 0.36 - 0.90 respectively). Similarly, those who stayed in admission for 6-12 days and for more than 12 days had a significant reduction in the odds of dying compared to those who stayed for less than 6 days (OR=0.20, 95%CI: 0.12 - 0.32 and OR=0.24, 95%CI: 0.15 - 0.37 respectively). Patients with two and more than two opportunistic infections had increased odds of dying compared to those with no opportunistic infection (OR=2.35, 95%CI: 1.35 - 4.06 and OR=4.23, 95%CI: 1.62 - 11.12 respectively). Finally, patients with a haemoglobin < 10g/l had increased odds of dying compared to those with higher levels (OR=2.38, 95%CI: 1.58 - 3.59) (Table 3).
| Factors | Total | AliveN (%) | Deceased N (%) | OR (95% CI) | P value | aOR (95% CI) | P value |
|---|---|---|---|---|---|---|---|
| Age, years | - | - | - | - | - | - | - |
| <37 | 301 | 235(78.1) | 66(21.9) | Ref | - | Ref | - |
| 37-48 | 309 | 237(76.7) | 72(23.3) | 1.08(0.74 – 1.58) | 0.64 | 1.05(0.67 – 1.65) | 0.10 |
| >48 | 281 | 210(74.7) | 71(25.3) | 1.20(0.82 – 1.77) | - | 1.58(1.00 – 2.49) | - |
| Sex | - | - | - | - | - | - | - |
| Men | 460 | 347(75.4) | 113(24.6) | Ref | - | - | - |
| Women | 431 | 335(77.7) | 96(22.3) | 0.87(0.64 – 1.20) | 0.42 | - | - |
| Calendar year | - | - | - | - | - | - | - |
| 2007-2009 | 210 | 150(71.4) | 60(28.6) | Ref | - | Ref | - |
| 2010-2012 | 308 | 249(80.8) | 59(19.2) | 0.59(0.39 – 0.89) | 0.04 | 0.53(0.33 – 0.87) | 0.02 |
| 2013-2015 | 373 | 283(75.9) | 90(24.1) | 0.79(0.54 – 1.17) | - | 0.57(0.36 – 0.90) | - |
| Hospital stay, days | - | - | - | - | - | - | - |
| <6 | 322 | 205(63.7) | 117(36.3) | Ref | - | Ref | - |
| 6-12 | 269 | 238(88.5) | 31(11.5) | 0.22(0.14 – 0.36) | <0.0001 | 0.20(0.12 – 0.32) | <0.0001 |
| >12 | 286 | 238(83.2) | 48(16.8) | 0.35(0.24 – 0.52) | - | 0.24(0.15 – 0.37) | - |
| Residence | - | - | - | - | - | - | - |
| Urban | 817 | 626(76.6) | 191(23.4) | Ref | - | - | - |
| Rural | 38 | 28(73.7) | 10(26.3) | 1.17(0.56 – 2.45) | 0.68 | - | - |
| Marital status | - | - | - | - | - | - | - |
| Married | 505 | 385(76.2) | 120(23.8) | Ref | - | - | - |
| Alone (single, widowed, divorced) | 347 | 267(76.9) | 80(23.1) | 0.96(0.70 – 1.33) | 0.81 | - | - |
| Employment | - | - | - | - | - | - | |
| Active | 642 | 493(76.8) | 149(23.2) | Ref | - | - | - |
| Unemployed | 194 | 145(74.7) | 49(25.3) | 1.12(0.77 – 1.62) | 0.81 | - | - |
| Retired | 41 | 32(78.1) | 9(21.9) | 0.93(0.43 – 2.00) | - | - | - |
| ARV | - | - | - | - | - | - | - |
| Yes | 531 | 416(78.3) | 115(21.7) | Ref | - | - | - |
| No | 333 | 247(74.2) | 86(25.8) | 1.26(0.91 – 1.74) | 0.16 | - | - |
| CD4, cells/mm3 | - | - | - | - | - | - | - |
| <200 | 476 | 368(77.3) | 108(22.7) | Ref | - | - | - |
| 200-349 | 102 | 84(82.4) | 18(17.6) | 0.73(0.42 – 1.7) | 0.12 | - | - |
| >350 | 81 | 70(86.4) | 11(13.6) | 0.54(0.27 – 1.05) | - | - | - |
| Opportunistic infections | - | - | - | - | - | - | - |
| None | 215 | 164(76.3) | 51(23.7) | Ref | - | Ref | - |
| 1 | 474 | 382(80.6) | 92(19.4) | 0.77(0.52 – 1.14) | - | 0.96(0.60 – 1.54) | - |
| 2 | 175 | 120(68.6) | 55(31.4) | 1.47(0.94 – 2.31) | 0.002 | 2.35(1.36 – 4.08) | 0.0001 |
| >2 | 27 | 16(59.3) | 11(40.7) | 2.21(0.96 – 5.11) | - | 4.25(1.62 – 11.14) | - |
| Haemoglobin, g/l | - | - | - | - | - | - | - |
| ≥10 | 291 | 247(84.9) | 44(15.1) | Ref | - | Ref | - |
| <10 | 494 | 362(73.3) | 132(26.7) | 2.05(1.40 – 3.00) | <0.0001 | 2.38(1.58 – 3.59) | <0.0001 |
4. DISCUSSION
We aimed at determining the trends, causes and factors associated with in-hospital mortality in HIV/AIDS patients in the Douala General Hospital. The overall all- cause mortality rate during the study period was 23.6%. The category - communicable diseases and AIDS defining malignancies was the leading cause of mortality. Tuberculosis was the most common specific cause of mortality (24.3%). Factors associated with in-hospital mortality were; length of hospital stay, number of opportunistic infections, and severity of anaemia.
In this setting, valid data on vital statistics such as mortality rates are scarce due to inefficient systems for national registration of events. Most studies which describe mortality patterns still rely mainly on hospital data [29]. These are prone to selection bias as patients admitted to the hospital may not be representative of all patients in the community and may not also be a true reflection of causes of deaths in the general population. They can nevertheless still give an insight into the burden of a particular disease at a specified period in time. Despite these limitations, in-hospital mortality can be an important indicator of a public health system’s response to the HIV epidemic thereby contributing to public health policy [30-32]
The overall all-cause mortality of 23.6% was lower than found in similar studies in SSA as in Nigeria, Ghana, Namibia and Senegal where it ranged from 31 to 38% [18-20, 22, 33] and 37.4% in Libya [34]. In a systematic review and meta-analysis, worldwide, in-hospital mortality among HIV-positive adults was 20% overall, but highest in the Africa region with 31% [23]. In two previous studies in Cameroon, in 2009 [35] and 2013 [36] mortality was 28.5% and 16.3% respectively. Their methodology was different. Both were retrospective cohorts, all patients were on cART, and followed up in the treatment centre as out-patients. High in-patient mortality calls for improvements in patient services through improved diagnosis and management, especially as the major causes of deaths are communicable conditions. Most often, diagnosis of these conditions is clinical and patients treated empirically [37]. The high death rates due to treatable and preventable AIDS associated conditions suggest that appropriate interventions focused on early HIV diagnosis with initiation of effective cART (less than 40% of our study population were on cART) and prophylaxis against OI, could avert a majority of deaths in our population [38].
There seemed to be two phases in the trends in mortality from our study which should be interpreted with caution because there was no significant change in trend. In the first phase from 2007 to 2011, we observed a downward trend. Combination ART was introduced free of charge in 2007 with awareness campaigns and increased scale up of treatment. The overall mortality decreased from 38% to 18%. This decline in mortality as was expected has been corroborated in other studies in SSA [17, 39]and also in resource rich countries where this is the usual trend [40-42]. We found an unexpected but sustained rise in mortality in the years 2012 to 2014. The potential reasons for this need to be evaluated. There is supposed to be improved access to care and thus better mortality indicators are expected. Considerations could include late presentation to HIV care, individual patient factors and low HAART uptake [43, 44]. This analysis was beyond the scope of this work. However, any rise in mortality is worrisome as it could degenerate to an eventual failure of this public health program. The drop in mortality in 2015 was therefore welcomed but unfortunately we did not study the reasons for this, while hoping it remains sustained.
The distribution of causes of deaths in different studies may not be directly comparable due to different patient characteristics such as gender, age- groups, socio-economic status and geographical region [23, 45]. The presence of over 70% of patients presenting with CD4 counts below 200, 75% presenting with opportunistic infections and only 38% on HAART can explain why the ‘’communicable conditions and AIDS defining malignancy category”” was still predominant. Unfortunately, the causes of death in this supposedly cART era in Cameroon, was similar to the pre and early cART era in Canada where overall 85% of all deaths were attributable to AIDS related conditions. Still in this study, there was a steady decline over the years to 67% six years later [46].
Of the specific causes of death, our findings of Tuberculosis to be the most common cause of death (one in four deaths) was similar to other SSA studies [17, 19-21, 23]. Tuberculosis is endemic in this setting and HIV infection is the strongest risk factor for developing tuberculosis which has fuelled its resurgence [38, 47]. HIV associated Central Nervous System disease (including mainly cryptococcal meningitis, toxoplasma encephalitis and other meningitis syndromes) being present in 23.4% of our study population was a close second behind TB. This increased prevalence and the high mortality associated with it had previously been reported in this setting [48] where 1 in 2 patients admitted died. Again, late presentation to HIV, lack of diagnostic and efficient management may all be associated with mortality.
The presence of OI and severe anaemia was independently associated with mortality. These have been previously described [16, 35, 36]. This probably reflects the advanced clinical stages at which patients present for care, mortality being highest among patients with severe immune-depression. Also, high mortality has been shown to be associated with multiple diagnosis. Multiple diagnosis predicts a poor outcome in HIV/AIDS and should be recognised early so that additional care and support of these individuals should be provided on admission [37].
5. LIMITATIONS
This was a retrospective review of patient records and thus prone to missing data. We did not have any rationale to apply multiple imputation techniques to estimate missing values, because the files were not conceived to answer our questions; thus, we considered that values were missing completely at random. Another cause of missing data could be due to the outcome of the patients as those who died early may have had less data. Despite the missing values, we still had a relatively big sample size to answer the addressed questions, even though some important variables were not considered for the analysis of predictors of mortality because almost half of the data on some variables were missing. Interpreting the trend in mortality over time must be treated with caution as there was the susceptibility of change of practice. For example, management guidelines for HIV changed over time during the study period. Ascertaining the underlying cause of death remained a very big challenge in this study because death certificates are not standard and most of the times may not be accurately filled. The underlying cause of death for most patients was ascertained years after the event had occurred as this information was not found in some of the files. The method used could be subject to misclassification bias. The Douala general hospital being a referral hospital, its hospitalised patients might be different from those from other hospitals and those in the general population, making the generalisation of our findings questionable. In spite of these limitations, we succeeded to show a clear picture of a relatively high mortality amongst hospitalised HIV/AIDS patients over time.
CONCLUSION
The overall all-cause mortality of hospitalized HIV/AIDS patients in the DGH was high as almost one in every four patients died. Generally, mortality decreased over time from 2007 as was consistent with other studies although there was a rise in the period 2012-2014 which needs to be further studied. The most common all-cause mortality was in the category “Communicable diseases and AIDS defining malignancy” of which tuberculosis was the most common specific cause of death. Factors shown to be associated with mortality were age, the year of hospitalisation, length of hospital stay, number of opportunistic infections, and anaemia. We hope that our findings will help to guide health policies aimed at reducing in-hospital mortality in HIV/AIDS.
LIST OF ABBREVIATIONS
| AIDS | = Acquired Immune Deficiency Syndrome; |
| AOR | = adjusted Odds Ratio; |
| cART | = combination Antiretroviral Therapy; |
| CI | = Confidence Interval; |
| HIV | = Human Immunodeficiency Virus; |
| ATC | = Accredited Treatment Centre, |
| IQR | = Interquartile Range; |
| OR | = Odds Ratio; |
| WHO | = World Health Organization; |
| DGH | = Douala General Hospital; |
| OI | = Opportunistic Infection; |
| SSA | = Sub-Saharan Africa. |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Ethical approval was obtained from the Institutional Review Board of the Faculty Medicine and Pharmaceutical Sciences of the University of Douala (IEC-UD/474/02/2016/T). Administrative approval was obtained from the Director of the Douala General Hospital.
HUMAN AND ANIMAL RIGHTS
No animals were used. All the reported experiments on human were in accordance with the National Aids Control Committee guidelines which were adapted from the WHO treatment guidelines.
CONSENT FOR PUBLICATION
Written and informed consent was obtained for the study.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
The study team would like to thank the DGH ATC members and staff of the medical wards for their support in facilitating this study.


