All published articles of this journal are available on ScienceDirect.
Correlation of High Interleukin 17A and Interleukin 6 Levels with High Virus Load Among Subtype C HIV-infected, Antiretroviral Therapy-naive Zimbabwean Patients: A Cross-sectional Study
Abstract
Introduction:
In response to the human immunodeficiency virus (HIV) infection, activated immune cells produce several cytokines that alter the immune response and HIV disease progression. We quantified Th1/Th2/Th17 cytokines in an antiretroviral therapy naïve (ART) cohort to investigate their correlation with traditional markers of HIV disease progression; CD4+ T-lymphocytes and virus load (VL).
Methods:
We enrolled 247 HIV-infected ART-naïve participants into the study. CD4+ T- and CD8+ T-lymphocytes were enumerated using flow cytometry. VL was quantified using the Cavidi ExaVirTM Load assay. IL-2, IL-4, IL-6, IL-10, IL-17A, TNF-α, and IFN-γ levels were quantified using the BD Cytometric Bead Array Human Th1/Th2/Th17 cytokine assay. The Kendall’s rank correlation coefficient was used to determine the correlation between log10 transformed data for cytokine levels and CD4+ T- and CD8+ T-lymphocytes, CD4/CD8 ratio, and VL.
Results:
The median CD4+ T- and CD8+ T-lymphocyte counts were 458 cells/µL (IQR:405-556) and 776 cells/µL (IQR:581-1064), respectively. The median CD4/CD8 ratio was 0.6 (IQR: 0.45-0.86). The median VL was log103.3.copies/mL (IQR:2.74-3.93). Low CD4+ T-lymphocyte counts (p=0.010) and CD4/CD8 ratio (p=0.044) were significantly correlated with high VL. There was no significant correlation of cytokine levels with CD4+ T-, CD8+ T-lymphocyte counts and CD4/CD8 ratio. However, high levels of IL-17A (p=0.012) and IL-6 (p=0.034) were significantly correlated with high VL.
Conclusion:
Our study contributes to the little knowledge available on the role of cytokine profiles in the immune response to subtype C HIV infection.
1. INTRODUCTION
Cytokines are crucial to cellular communication and thus may be key elements in turning on and off the immune response [1]. They play a critical role in immune system homeostasis.
Following HIV infection, immune cells are activated, proliferate and differentiate into effector cells which secrete various cytokines. There is an intense cytokine “storm” during acute HIV infection [2] which might contribute to severe immune activation, rapid CD4+ T-lymphocyte depletion and increase in virus replication leading to high virus load. Although CD4+ T-lymphocytes and virus load are considered as traditional markers of HIV disease progression [3], changes in the cytokine signature directly affect HIV disease progression [4]. Interleukin-6 (IL-6) has been reported to potentially increase virus replication [5, 6] while interferon (IFN)-α and IFN-β inhibit HIV replication [6, 7]. Other cytokines; Transforming Growth Factor (TGF)-β, IL-10, IL-13, and IL-4 have been shown to have both activating and inhibitory roles on HIV replication [7].
The balance of Th1 and Th2 cytokines has long been postulated to contribute to HIV disease progression [8, 9]. The Th1 to Th2 cytokine shift in HIV immunopathology remains controversial [10]. In the current study, we measured cytokine concentrations of IL-2, IL-4, IL-6, IL-10, Tumour Necrosis (TNF)-α, IFN-γ, and IL-17A to determine their correlation if any, with CD4+ T-lymphocyte counts and virus load, the traditional biomarkers of HIV disease progression. Additionally, we also investigated the correlation of CD8+ T-lymphocyte count, important in defence against intracellular pathogens and CD4/CD8 ratio with virus load and cytokine levels.
2. METHODS
2.1. Study participants
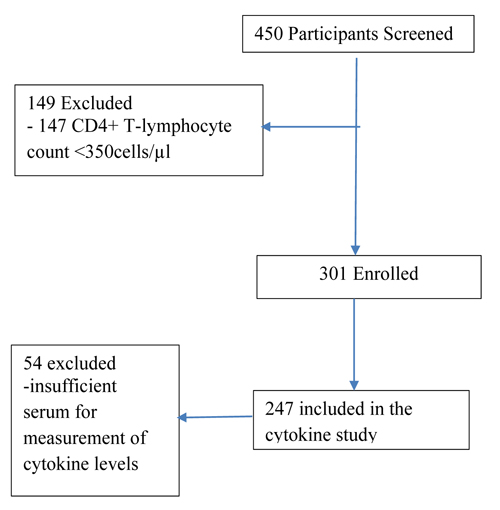
As summarised in Fig. (1), 450 potential study participants were screened based on their CD4+ T-lymphocyte count. Two hundred and forty seven (247) HIV-infected participants aged ≥ 18 years with a CD4+ T-lymphocyte count ≥ 350 cells/μL were enrolled into the “Virological and immunological investigations of HIV-infected individuals in Harare, Zimbabwe, a prospective study”, conducted at Parirenyatwa Central Hospital Opportunistic Infections Clinic, Harare, Zimbabwe between 2010 and 2013. During this period, the Zimbabwe Ministry of Health under the Zimbabwe National ART program was providing ART to HIV-infected patients with CD4+ T-lymphocyte count <350 cells/μL, based on the WHO recommendations. Prospective participants with active Tuberculosis were excluded from the study irrespective of their CD4+ T-lymphocyte count. Written informed consent was obtained from each participant. Demographic and medical history were collected at enrolment. The study was approved by the Medical Research Council of Zimbabwe (MRCZ/A/1558).
2.2. Enumeration of T-lymphocytes, CD4/CD8 Ratio and Virus Load Quantification
At enrolment, whole blood collected in EDTA tubes was used for enumeration of absolute CD4+ T- and CD8+ T-lymphocytes within 6 hours of blood collection using a Partec Cyflow counter (Cyflow, Partec, Munster, Germany) as previously described [11, 12].
The remaining whole blood was centrifuged and plasma was aliquoted and stored at -80°C until assayed. Virus load quantification was done using the Cavidi ExaVirTM Load version 3 assay (Cavidi AB, Uppsala Science Park, Sweden) following the manufacturer’s instructions [13, 14]. The assay measures the HIV reverse transcriptase activity in patient’s plasma as a marker for virus replication.
2.3. Measurements of Serum Cytokine Levels
Serum was collected at enrolment and stored at -80oC until further use. We used the BD Cytometric Bead Array (CBA) Human Th1/Th2/Th17 Cytokine assays (Becton, Dickson Company, BD Biosciences, CA 95131 USA.) which simultaneous quantification of seven cytokines (IL-2, IL-4, IL6, IL-10, TNF-α, IFN-γ, and IL-17A) following the manufacturer’s instructions.
2.4. Statistical Analysis
The Kendall’s rank correlation coefficient (Ƭ) was used to determine the correlation between log10 transformed data for cytokine levels and CD4+ T- and CD8+ T-lymphocytes, CD4/CD8 ratio, and virus load. All analysis was done using STATA version 13.1 (StataCorp, Texas, USA) and Graph Pad Prism7 (GraphPad Software Inc, California, USA).
3. RESULTS
3.1. Demographics of the Study Population
A total of 450 prospective study participants were screened. Three hundred and one (301) participants with CD4+ T lymphocyte count greater than 350 cells/µL were enrolled the study. Two hundred and forty-seven (247/301), 82% who had sufficient archived serum specimens were included in the current study (Fig. 1).
3.2. T-lymphocytes Counts and Virus Load
The median CD4+ T- and CD8+ T-lymphocyte counts were 458 cells/μL, inter quartile range (IQR): 405-556 cells/μL and 776 cells/μL (IQR:581-1064), respectively, whilst the median CD4/CD8 ratio was 0.6 (IQR:0.45-0.86). The median virus load was 3.3 Log10 copies/mL, IQR (2.74-3.93) (Table 1).
| T-lymphocytes | Viral Load | |
|---|---|---|
| Ƭ | p-value | |
| CD4+ T-lymphocytes | -0.115 | 0.010* |
| CD8+ T-lymphocytes | 0.044 | 0.328 |
| CD4/CD8 ratio | -0.091 | 0.044* |
| Cytokine | CD4+T-lymphocytes | Viral load | CD8+T-lymphocytes | CD4/CD8 ratio | ||||
|---|---|---|---|---|---|---|---|---|
| Ƭ | p-value | Ƭ | p-value | Ƭ | p-value | Ƭ | p-value | |
| IL-17A | 0.0545 | 0.214 | -0.1107 | 0.012* | 0.037 | 0.397 | -0.028 | 0.551 |
| IFN-γ | 0.0065 | 0.870 | 0.0578 | 0.142 | 0.148 | 0.221 | -0.068 | 0.177 |
| TNF-α | 0.0318 | 0.429 | -0.0515 | 0.199 | 0.018 | 0.649 | -0.005 | 0.926 |
| IL-10 | -0.0290 | 0.505 | -0.0484 | 0.265 | 0.011 | 0.798 | -0.024 | 0.616 |
| IL-6 | -0.0503 | 0.263 | -0.0955 | 0.034* | 0.057 | 0.202 | -0.074 | 0.104 |
| IL-4 | -0.0076 | 0.833 | 0.0044 | 0.905 | -0.018 | 0.619 | 0.008 | 0.877 |
| IL-2 | 0.0204 | 0.593 | -0.0665 | 0.081 | -0.019 | 0.605 | 0.028 | 0.575 |
3.3. Correlation of CD4+ T-, CD8+ T-lymphocyte Counts, CD4/CD8 Ratio with Viral Load
Low CD4+ T-lymphocyte counts (Ƭ =-0.115, p=0.010) and CD4/CD8 ratio (Ƭ =-0.091, p=0.044) were correlated with high viral load whilst there was no significant correlation between CD8+ T-lymphocyte counts with viral load (Ƭ =0.044, p=0.328), (Table 1).
The Kendall’s rank correlation coefficient (Ƭ) was used to determine the correlation between CD4+ T- and CD8+ T-lymphocytes, CD4/CD8 ratio with virus load. *P-value <0.05 was considered statistically significant.
3.4. Correlation of Cytokines Levels with CD4+ T-, CD8+ T-lymphocyte Counts, CD4/CD8 Ratio and Virus Load
There was no significant correlation between cytokine levels and CD4+ T-, CD8+ T-lymphocyte counts and CD4/CD8 ratio (Table 2 and Fig. 2). However, high concentrations of IL-17A (Ƭ =-0.1107, p=0.012) and IL-6 (Ƭ=-0.0955, p=0.034) were significantly correlated with high virus load.
The Kendall’s rank correlation coefficient (Ƭ) was used to determine the correlation between log10 transformed data for cytokine levels and CD4+ T- and CD8+ T-lymphocytes, CD4/CD8 ratio and virus load.*P-value <0.05 was considered statistically significant.
Based on the medical history collected at enrolment, the median time since the diagnosis of HIV infection was 6.3 years, thus the majority of the participants can be considered as chronically infected.
4. DISCUSSION
Cytokine levels in an ART naïve cohort infected predominantly with HIV subtype C the major subtype in Zimbabwe [15] were measured to investigate their correlation with CD4+ T-lymphocyte counts and virus load, the traditional biomarkers of HIV disease progression. Based on Kendall’s rank correlation, high IL-17A and IL-6 levels were correlated with high viral load. Interestingly, none of the measured cytokines were significantly correlated with CD4+ T- and CD8+ T-lymphocytes counts or CD4/CD8 ratio. However, we confirmed the inverse correlation of low CD+T-lymphocyte counts with high virus load as has been established in previous studies [3]. We also noted that although CD8+ T-lymphocyte counts were not correlated with viral load, interestingly, low CD4/CD8 ratio was correlated with high virus load and thus may be used as a potential marker of HIV disease progression. It is well established that whilst there is a depletion of CD4+ T-lymphocytes counts in HIV infection, there is a compensatory increase in CD8+T-lymphocyte leading to a reduced CD4/CD8 ratio which is normally greater than 1 [16]. The role of CD8+ T-lymphocyte counts in immune response to HIV infections is not fully understood as opposed to its well-studied functional capacity. In a previous study [17] we showed that the CD4/CD8 ratio could be used as a potential tool for diagnosis of HIV infection in infants under the age of 18 months and indeed in adults.
IL-17A is a major inflammatory mediator and IL-17A producing CD4+ T-lymphocytes (Th17) are a key subset of cells maintaining mucosal integrity and host defence against pathogens through mucosal barrier [16, 18]. IL-17A may induce production of IL-1β and IL-6 pro-inflammatory cytokines [19] which are strongly linked to inflammatory disorders [20]. Although the current study did not measure mucosal cytokine levels, there is strong evidence of a critical role mucosal immunity plays in HIV transmission [21, 22]. The proposed broken mucosal surfaces imply a weakened immune system which promotes HIV transmission in the mucosal tissue. Mucosal cytokine levels have been used as markers of the effectiveness of microbicides aimed at blocking viral transmission [23]. Th17 are suggested to favour viral replication during acute HIV infection [24, 25], and accelerated disease progression in chronically infected individuals through inefficient mucosal repair, increased microbial translocation and hyperactivation of the immune system [26, 27]. These findings may plausibly explain the correlation of IL-17A levels with virus load in the current study.
In addition to IL17, IL-6 levels were significantly correlated with virus load in the current study. IL-6 mediates Th17 generation [28, 29] which promotes a pro-inflammatory status and contribute to inflammation through induction of acute-phase proteins like C-reactive protein [30]. IL-6 has been reported to enhance HIV replication at the transcriptional and post-transcriptional level [6]. It seems the correlation of IL-6 levels with virus load in our study may be explained by the inflammatory environment promoted by IL-6. Generally, inflammation-inducing cytokines (for example IL-17A and IL-6) promote HIV replication coupled with CD4+ T-lymphocyte count decline through enhanced proviral transcription, recruitment, and activation of CD4+ T-lymphocyte, and activation-induced apoptosis by bystander T-lymphocytes [31-33].

Although in our study the CD4+ T-lymphocyte counts and virus load did not correlate with IL-2, IL-4, and IFN-γ, others have reported that these cytokines are needed for activation of antiviral T-lymphocyte expansion and humoral immunity which may then increase viral replication through activation of HIV-infected cells [31]. During HIV infection, high levels of inflammatory cytokines (IL-12p40, IL-12p70, IFN-γ, IL-7, and IL-15) are associated with high viraemia and a decrease in CD4+ T-lymphocyte counts [34]. In late-stage HIV infection, low levels of IL-2 and IFN-γ plus elevated levels of IL-4 and IL-10 are associated with a poor HIV antigen-specific immunity coupled with emergence of opportunistic infections [11]. Additionally, IL-12p40 has been reported to be a good biomarker for CD4+ T-lymphocyte decline and virus load increase in subtype C infection [35]. Conversely, Twizerimana et al. [36] demonstrated IL-10 down-regulation of Th1 cytokines and HIV disease progression associated Th1 to Th2 cytokine shift.
HIV-1 infection is characterized by a marked depletion of CD4+ T-lymphocytes especially in gut-associated lymphoid tissues coupled with immune hyperactivation through microbial translocation [37]. Although we speculate that the correlation of IL-17A and IL-6 levels with virus load is through impaired mucosal immunity and continuous immune hyperactivation, one of the limits of our study is that we did not investigate the source of these cytokines. Additional study limits include the cross-sectional nature of our study that did not allow repeated measurements of cytokines during the course of participants’ follow-up to correlate with changes of CD4+ T-lymphocyte counts and virus load. Repeated cytokine quantification over time would shed more light on the potential of cytokine profiles as biomarkers of HIV disease progression.
CONCLUSION
Low CD4+ T- lymphocytes and CD4/CD8 ratio were significantly correlated with high VL. Whilst CD4+ and CD8+T-lymphocytes counts were not correlated with cytokine levels, high levels of IL-17A and IL-6 were significantly correlated with VL. Our study contributes to the growing knowledge the role of cytokine profiles in the immune response to HIV infection.
LIST OF ABBREVIATIONS
| ART | = Antiretroviral Therapy |
| CD4 | = Cluster of Differentiation 4 |
| CD8 | = Cluster of Differentiation 8 |
| CI | = Confidence Interval |
| IQR | = Interquartile Range |
| HIV | = Human Immunodeficiency Virus |
| IFN | = Interferon |
| IL | = Interleukin |
| MRCZ | = Medical Research Council of Zimbabwe |
| Th | = T helper |
| TNF | = Tumour Necrosis Factor |
| TGF | = Transforming Growth Factor |
| VL | = Virus Load |
AUTHORS’ CONTRIBUTIONS
TM and LSZ conceived and designed the study. TM, MT, and BM performed the assays. TB and KM conducted the data analysis. TM wrote the first draft.
All authors reviewed and approved the final version of the manuscript.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was approved by the Medical Research Council of Zimbabwe (MRCZ/A/1558).
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures followed were in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Written informed consent was obtained from each participant.
AVAILABILITY OF DATA AND MATERIALS
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
FUNDING
The study was funded by EDCTP I; TESA I: CG_cb_07_41700 and 1051 TESA II EDCTP-RegNet 2015 part of the EDCTP2 programme (supported under Horizon 2020, the European Union’s Framework Programme for Research and Innovation).
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We acknowledge data entry personnel Brian Nyauzame and Tambudzai Margaret Pswarayi. We thank the study participants for their participation in the study and the Parirenyatwa Opportunistic Infections Clinic staff.


